LONDON, UK: Though teething is a difficult experience for both parents and their babies, a judicious decision-making process should be employed when selecting a product to relieve it. This, at least, is the conclusion reached in a recent study regarding teething products in the UK.
The research examined all 14 teething products currently licensed for commercial sale in the UK by the Medicines and Healthcare products Regulatory Agency (MHRA). The results revealed that two products contained sucrose, which could cause newly erupted primary teeth to undergo decay, as these products are applied directly to the teeth. Six products were found to contain alcohol, which has a number of potential side effects, including impaired motor development.
All six of the teething gels licensed by MHRA were found to contain lidocaine at a 1% or less solution. In 2014, the US Food and Drug Administration found that the presence of viscous lidocaine at a 2% solution had been associated with 22 adverse reactions, including six deaths.
In response to the research, the British Dental Association (BDA) has urged parents to be aware of which teething products could be potentially harmful for their children.
“Parents buying teething powders to save infants from distress won’t always realise they’re offering their kids sugars, alcohol or lidocaine,” said Chair of the BDA’s Principal Executive Committee Dr Mick Armstrong.
“Buying a licensed product should offer confidence you’re making a safe choice. The reality is consumers are navigating a minefield of potentially harmful ingredients. We need to see real change in the way these products are licensed and marketed, and clear guidance so parents understand the risks. If your little one is suffering, then a teething ring kept cool in the fridge is all you need,” Armstrong added.
The study, titled “Teething products may be harmful to health”, was published on 27 September 2019 in the British Dental Journal.
Tags:
NEW YORK, U.S.: Many people enjoy a drink, and now researchers from the New York University (NYU) School of Medicine have identified that alcohol ...
BOSTON, U.S.: Root canal therapy and the materials and techniques used to perform them are continually evolving. However, the fact remains that after the ...
STOCKHOLM, Sweden: Understanding of cell types and the mechanisms of dental growth is essential for the reconstruction and engineering of teeth. Therefore, ...
PHILADELPHIA, U.S.: Taste disorders and other neurosensory defects have been reported postoperatively in a number of patients who have undergone extraction ...
VLADIVOSTOK, Russia: A group of histologists and dentists from the Far Eastern Federal University (FEFU) have collaborated with Russian and Japanese ...
PORTLAND, Ore., USA: Researchers at the Oregon Health and Science University (OHSU) have developed a revolutionary process by which to engineer new blood ...
Although orthodontic treatment has become increasingly commonplace, its periodontal side effects are often overlooked. In a free CURADEN webinar on Tuesday,...
COLUMBUS, Ohio, USA: As previous laboratory studies have suggested that dietary administration of whole strawberries has substantial potential as a strategy...
NEW PLYMOUTH, New Zealand: With its good weather and outdoor-focused style of life, it seems unexpected that obesity would be an issue in New Zealand. ...
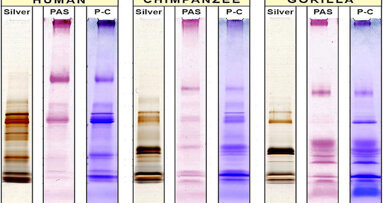
BUFFALO, N.Y., U.S.: Much is already known about saliva’s benefits for food digestion and general oral health. The differences between human saliva and ...
Live webinar
Tue. 26 May 2026
12:00 pm EST (New York)
Live webinar
Tue. 26 May 2026
2:00 pm EST (New York)
Live webinar
Wed. 27 May 2026
8:00 am EST (New York)
Live webinar
Thu. 28 May 2026
12:00 pm EST (New York)
Dr. Anthony Viazis DDS, MS. Inventor & Founder of Fastbraces®
Live webinar
Mon. 1 June 2026
1:00 pm EST (New York)
Live webinar
Wed. 3 June 2026
1:00 pm EST (New York)
Live webinar
Thu. 4 June 2026
2:00 pm EST (New York)



 Austria / Österreich
Austria / Österreich
 Bosnia and Herzegovina / Босна и Херцеговина
Bosnia and Herzegovina / Босна и Херцеговина
 Bulgaria / България
Bulgaria / България
 Croatia / Hrvatska
Croatia / Hrvatska
 Czech Republic & Slovakia / Česká republika & Slovensko
Czech Republic & Slovakia / Česká republika & Slovensko
 France / France
France / France
 Germany / Deutschland
Germany / Deutschland
 Greece / ΕΛΛΑΔΑ
Greece / ΕΛΛΑΔΑ
 Hungary / Hungary
Hungary / Hungary
 Italy / Italia
Italy / Italia
 Netherlands / Nederland
Netherlands / Nederland
 Nordic / Nordic
Nordic / Nordic
 Poland / Polska
Poland / Polska
 Portugal / Portugal
Portugal / Portugal
 Romania & Moldova / România & Moldova
Romania & Moldova / România & Moldova
 Slovenia / Slovenija
Slovenia / Slovenija
 Serbia & Montenegro / Србија и Црна Гора
Serbia & Montenegro / Србија и Црна Гора
 Spain / España
Spain / España
 Switzerland / Schweiz
Switzerland / Schweiz
 Turkey / Türkiye
Turkey / Türkiye
 UK & Ireland / UK & Ireland
UK & Ireland / UK & Ireland
 Brazil / Brasil
Brazil / Brasil
 Canada / Canada
Canada / Canada
 Latin America / Latinoamérica
Latin America / Latinoamérica
 USA / USA
USA / USA
 China / 中国
China / 中国
 India / भारत गणराज्य
India / भारत गणराज्य
 Pakistan / Pākistān
Pakistan / Pākistān
 Vietnam / Việt Nam
Vietnam / Việt Nam
 ASEAN / ASEAN
ASEAN / ASEAN
 Israel / מְדִינַת יִשְׂרָאֵל
Israel / מְדִינַת יִשְׂרָאֵל
 Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
 Middle East / Middle East
Middle East / Middle East




































To post a reply please login or register