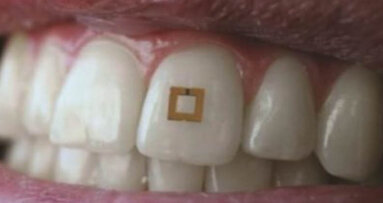
COLLEGE STATION, Texas, US: A new intra-oral biosensor developed in the US could transform how periodontal disease is detected and monitored. Aiming to help shift dentistry toward proactive disease monitoring, researchers at Texas A&M University in College Station have developed an intra-oral patch capable of detecting inflammatory biomarkers. Following successful testing, they say that the device establishes feasibility of effective point-of-care monitoring in the challenging oral cavity environment.
The biosensor is contained in a wearable patch and designed to continuously monitor the levels of target biomarkers within the oral cavity. Adhering directly to oral soft tissue, it targets the tumour necrosis factor alpha protein associated with inflammation and oral disease progression.
The performance of the biosensor was evaluated through in vitro, ex vivo and in vivo tests, which demonstrated effective adhesion, stable function in simulated dynamic oral conditions, and high sensitivity in detecting and quantifying the targeted protein. Study results demonstrated detection limits far below those typically detectable with conventional diagnostic methods.
Co-developer of the device Dr Chenglin Wu, associate professor in the Zachry Department of Civil and Environmental Engineering, explained in a university press release that the biosensor’s high sensitivity is achieved via a sensor surface engineered to detect very small amounts of the target protein, while a filtering layer helps prevent interference from other molecules in the oral environment. Dr Wu said: “For context, a patient with a viral infection might show symptoms at ten million or one billion virus copies per millilitre. Our sensor could detect 100 to 150 per millilitre.”
This sensitivity is supported by a hydrogel that provides robust adhesion. Dr Shaoting Lin, assistant professor at the Michigan State University College of Engineering, who collaborated with Dr Wu in developing the sensor, said in the press release: “Sensing measurements can be significantly influenced by the dynamic movement of tissues. A more robust tissue bond allows for a more reliable sensing performance independent of the strain.”
In their study, the researchers summarised: “Collectively, these findings establish the feasibility of real time in situ monitoring of inflammatory cytokines in the oral cavity and underscore the sensor’s potential for [point-of-care] diagnostics and personalised healthcare. Looking ahead, we aim to evaluate its clinical efficacy through expanded trials further, advancing its integration into routine dental practice and broader community health monitoring platforms.”
Although further validation in human clinical trials is required, the findings suggest broad potential applications. If successfully translated into clinical practice, such biosensors could significantly improve treatment outcomes, reduce healthcare costs and enhance patient engagement in preventive oral healthcare.
The article, titled “Tissue-adhesive hydrogel–MXene biosensor for in situ intraoral TNF-α detection”, was published online in the 16 January 2026 issue of Science Advances.
Topics:
Tags:
MEDFORD, Mass., US: Stress can present as a wide range of physical, emotional and social symptoms, such as irritability, headache, anxiety and elevated ...
MANCHESTER, England: Many dental professionals use behavioural techniques alongside routine oral hygiene advice to help patients improve their brushing and ...
GOTHENBURG, Sweden: As the popularity of snus continues to grow, so too does the diversity of products available, which now include portion-packed snus and ...
CAMBRIDGE, Mass., US: Most wearable electronics are worn on the skin, and intra-oral devices are rare. Researchers at Massachusetts Institute of ...
SOMERVILLE, Mass., U.S.: The dramatic shift toward more personalized dental treatment is perhaps no more evident than within digital dentistry. In another ...
What if we could replace the cumbersome process of validation for our devices, banking and other secure entry points with a flick of the tongue? According ...
GUANGZHOU, China: One of the keys to controlling dental caries and periodontal disease is early detection of dental lesions and cavities. A novel mouth ...
MASSACHUSETTS, U.S.: The digital world allows for a broad scope of data to be collected within a range of fields. In a new data collection advancement that ...
Live webinar
Thu. 2 April 2026
12:00 pm EST (New York)
Live webinar
Wed. 8 April 2026
1:00 pm EST (New York)
Live webinar
Thu. 9 April 2026
1:00 pm EST (New York)
Live webinar
Thu. 9 April 2026
2:00 pm EST (New York)
Prof. Moritz Kebschull, Cat Edney
Live webinar
Fri. 10 April 2026
10:00 am EST (New York)
Live webinar
Fri. 10 April 2026
11:00 am EST (New York)
Dr. med. dent. Henrik-Christian Carl Hollay
Live webinar
Fri. 10 April 2026
12:00 pm EST (New York)
Prof. Dr. Ali Murat Kökat



 Austria / Österreich
Austria / Österreich
 Bosnia and Herzegovina / Босна и Херцеговина
Bosnia and Herzegovina / Босна и Херцеговина
 Bulgaria / България
Bulgaria / България
 Croatia / Hrvatska
Croatia / Hrvatska
 Czech Republic & Slovakia / Česká republika & Slovensko
Czech Republic & Slovakia / Česká republika & Slovensko
 France / France
France / France
 Germany / Deutschland
Germany / Deutschland
 Greece / ΕΛΛΑΔΑ
Greece / ΕΛΛΑΔΑ
 Hungary / Hungary
Hungary / Hungary
 Italy / Italia
Italy / Italia
 Netherlands / Nederland
Netherlands / Nederland
 Nordic / Nordic
Nordic / Nordic
 Poland / Polska
Poland / Polska
 Portugal / Portugal
Portugal / Portugal
 Romania & Moldova / România & Moldova
Romania & Moldova / România & Moldova
 Slovenia / Slovenija
Slovenia / Slovenija
 Serbia & Montenegro / Србија и Црна Гора
Serbia & Montenegro / Србија и Црна Гора
 Spain / España
Spain / España
 Switzerland / Schweiz
Switzerland / Schweiz
 Turkey / Türkiye
Turkey / Türkiye
 UK & Ireland / UK & Ireland
UK & Ireland / UK & Ireland
 Brazil / Brasil
Brazil / Brasil
 Canada / Canada
Canada / Canada
 Latin America / Latinoamérica
Latin America / Latinoamérica
 USA / USA
USA / USA
 China / 中国
China / 中国
 India / भारत गणराज्य
India / भारत गणराज्य
 Pakistan / Pākistān
Pakistan / Pākistān
 Vietnam / Việt Nam
Vietnam / Việt Nam
 ASEAN / ASEAN
ASEAN / ASEAN
 Israel / מְדִינַת יִשְׂרָאֵל
Israel / מְדִינַת יִשְׂרָאֵל
 Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
 Middle East / Middle East
Middle East / Middle East





























To post a reply please login or register