The human body contains over 200 different types of cells, which are organised into tissues and organs that perform all the tasks required to maintain the viability of the system, including reproduction. In healthy adult tissues, the cell population size is the result of a fine balance between cell proliferation, differentiation, and death. Following tissue injury, cell proliferation begins to repair the damage. In order to achieve this, quiescent cells (dormant cells) in the tissue become proliferative, or stem cells are activated and differentiate into the appropriate cell type needed to repair the damaged tissue. Research into stem cells seeks to understand tissue maintenance and repair in adulthood and the derivation of the significant number of cell types from human embryos.
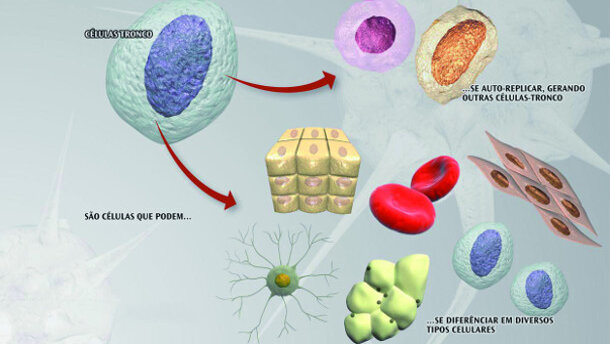
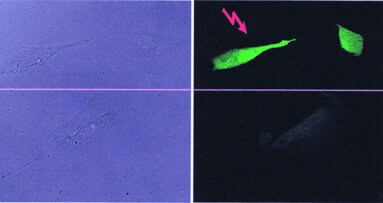
It has long been observed that tissues can differentiate into a wide variety of cells, and in the case of blood, skin and the gastric lining the differentiated cells possess a short half-life and are incapable of renewing themselves. This has led to the idea that some tissues may be maintained by stem cells, which are defined as cells with enormous renewal capacity (self-replication) and the ability to generate daughter cells with the capacity of differentiation. Such cells, also known as adult stem cells, will only produce the appropriate cell lines for the tissues in which they reside (Fig. 1).
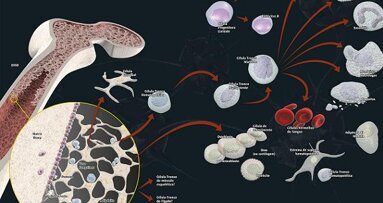
Not only can stem cells be isolated from both adult and embryo tissues; they can also be kept in cultures as undifferentiated cells. Embryo stem cells have the ability to produce all the differentiated cells of an adult. Their potential can therefore be extended beyond the conventional mesodermal lineage to include differentiation into liver, kidney, muscle, skin, cardiac, and nerve cells (Fig. 2).
embedImagecenter("Imagecenter_1_1145",1145, "large");
The recognition of stem cell potential unearthed a new age in medicine: the age of regenerative medicine. It has made it possible to consider the regeneration of damaged tissue or an organ that would otherwise be lost. Because the use of embryo stem cells raises ethical issues for obvious reasons, most scientific studies focus on the applications of adult stem cells. Adult stem cells are not considered as versatile as embryo stem cells because they are widely regarded as multipotent, that is, capable of giving rise to certain types of specific cells/tissues only, whereas the embryo stem cells can differentiate into any types of cells/tissues. Advances in scientific research have determined that some tissues have greater difficulty regenerating, such as the nervous tissue, whereas bone and blood, for instance, are considered more suitable for stem cell therapy.
In dentistry, pulp from primary teeth has been thoroughly investigated as a potential source of stem cells with promising results. However, the regeneration of an entire tooth, known as third dentition, is a highly complex process, which despite some promising results with animals remains very far from clinical applicability. The opposite has been observed in the area of jawbone regeneration, where there is a higher level of scientific evidence for its clinical applications. Currently, adult stem cells have been harvested from bone marrow and fat, among other tissues.
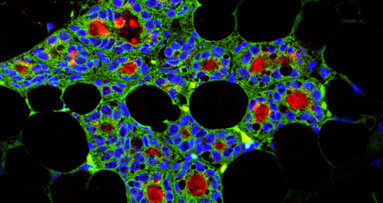
Bone marrow is haematopoietic, that is, capable of producing all the blood cells. Since the 1950s, when Nobel Prize winner Dr E. Donnall Thomas demonstrated the viability of bone marrow transplants in patients with leukaemia, many lives have been saved using this approach for a variety of immunological and haematopoietic illnesses. However, the bone marrow contains more than just haematopoietic stem cells (which give rise to red and white blood cells, as well as platelets, for example); it is also home to mesenchymalstem cells (which will become bone, muscle and fat tissues, for instance; Fig. 3).
Bone marrow harvesting is carried out under local anaesthesia using an aspiration needle through the iliac (pelvic) bone. Other than requiring a competent doctor to perform such a task, it is not regarded as an excessively invasive or complex procedure. It is also not associated with high levels of discomfort either intra or post-operatively (Figs. 4a & b).
Bone reconstruction is a challenge in dentistry (also in orthopaedics and oncology) because rebuilding bony defects caused by trauma, infections, tumours or dental extractions requires bone grafting. The lack of bone in the jaws may impede the placement of dental implants, thus adversely affecting patients’ quality of life. In order to remedy bone scarcity, a bone graft is conventionally harvested from the chin region or the angle of the mandible. If the amount required is too large, bone from the skull, legs or pelvis may be used. Unlike the process for harvesting bone marrow, the process involved in obtaining larger bone grafts is often associated with high levels of discomfort and, occasionally, inevitable post-operative sequelae (Figs. 5a–e).
The problems related to bone grafting have encouraged the use of bone substitutes (synthetic materials and bone from human or bovine donors, for example). However, such materials show inferior results compared with autologous bone grafts (from the patient himself/herself), since they lack autologous proteins. Therefore, in critical bony defects, that is, those requiring specific therapy to recover their original contour, a novel concept to avoid autologous grafting, involving the use of bone-sparing material combined with stem cells from the same patient, has been gaining ground as a more modern philosophy of treatment. Consequently, to the detriment of traditional bone grafting (with all its inherent problems), this novel method of combining stem cells with mineralized materials uses a viable graft with cells from the patient himself/herself without the need for surgical bone harvesting.
Until recently, no studies had compared the different methods available for using bone marrow stem cells for bone reconstruction. In the fol lowing paragraphs, I shall summarise a study conducted by our research team, which entailed the creation of critical bony defects in rabbits and subsequently applying each of the four main stem cell methods used globally in order to compare their effectiveness in terms of bone healing:(1)
- fresh bone marrow (without any kind of processing);
- a bone marrow stem cell concentrate;
- a bone marrow stem cell culture; and
- a fat stem cell culture (Figs. 6 & 7).
In a fifth group of animals, no cell therapy method (control group) was used. The best bone regeneration results were found in the groups in which a bone marrow stem cell concentrate and a bone marrow stem cell culture were used, and the control group showed the worst results. Consequently, it was suggested that stem cells from bone marrow would be more suitable than those from fat tissue for bone reconstruction and that a simple stem cell concentrate method (which takes a few hours) would achieve similar results to those obtained using complex cell culture procedures (which take on average three to four weeks; Figs. 8a & b).
Similar studies performed in humans have corroborated the finding that bone marrow stem cells improve the repair of bony defects caused by trauma, dental extractions or tumours. The histological images below illustrate the potential of bone-sparing materials combined with stem cells for bone reconstruction (Fig. 9). It is clear that the level of mineralised tissue is significantly higher in those areas where stem cells were applied (Figs. 10a & b).
Evidently, although bone marrow stem cell techniques for bone reconstruction are very close to routine clinical use, much caution must be exercised before indicating such a procedure. This procedure requires an appropriately trained surgical and laboratory team, as well as the availability of the necessary resources (Figs. 11a–h, taken during laboratory manipulation of marrow stem cells at São Leopoldo Mandic dental school in Brazil).
1. André Antonio Pelegrine, Antonio Carlos Aloise, Allan Zimmermann et al., Repair of critical-size bone defects using bone marrow stromal cells: A histomorphometric study in rabbit calvaria. Part I: Use of fresh bone marrow or bone marrow mononuclear fraction, Clinical Oral Implants Research, 00 (2013): 1–6.
2. André Antonio Pelegrine, Antonio Carlos Aloise & Carlos Eduardo Sorgi da Costa, Células Tronco em Implantodontia (São Paulo: Napoleão, 2013).
This article was published in cosmetic dentistry No. 04/2013
The human body contains over 200 different types of cells, which are organised into tissues and organs that perform all the tasks required to maintain the ...
ZURICH, Switzerland: The ability of adult stem cells to generate various tissue-specific cell populations allows for the replacement of damaged cells and ...
RIVERSIDE, Calif., U.S.: In the next few years, dentists may start to see the effects of electronic cigarettes (ECs) in more detail. Promoted as a safer ...
Material and methods
Cells
Human stem cells from the dental pulp of adults (given by S. Gronthos, NIH, Bethesda, USA) were cultivated in α-modified ...
Today, about 65 % of Italian dentists are prac tising implantology. In Italy alone, over a million implants are placed every year. A survey commissioned by ...
PHILADELPHIA, U.S.: Stem cell research has been central to many developments in medicine. In a new study, scientists have investigated whether stem cells ...
KILCHBERG, Switzerland: Ceramic implants are increasingly being used in modern dental implantology as a complement to the treatment spectrum with titanium ...
As part of its focus on clinical education, Dentsply Sirona will feature two new webinars on its clinical educational platform—Dentsply Sirona Academy ...
BASEL, Switzerland/HELSINKI, Finland: Haematopoietic stem cell transplantation is used to treat cancers and severe blood and autoimmune diseases. Owing to ...
ZURICH, Switzerland: Global implant dentistry company Nobel Biocare has announced the first recipients of its newly established Medal of Excellence in ...
Live webinar
Wed. 3 June 2026
1:00 pm EST (New York)
Live webinar
Thu. 4 June 2026
2:00 pm EST (New York)
Live webinar
Mon. 8 June 2026
12:00 pm EST (New York)
Live webinar
Mon. 8 June 2026
1:00 pm EST (New York)
Dr. Anthony Mak B.D.S, Prof. Marleen Peumans
Live webinar
Mon. 8 June 2026
2:00 pm EST (New York)
Live webinar
Wed. 10 June 2026
11:00 am EST (New York)
Live webinar
Wed. 10 June 2026
2:00 pm EST (New York)
Nacho Fernández-Baca DDS, MSc



 Austria / Österreich
Austria / Österreich
 Bosnia and Herzegovina / Босна и Херцеговина
Bosnia and Herzegovina / Босна и Херцеговина
 Bulgaria / България
Bulgaria / България
 Croatia / Hrvatska
Croatia / Hrvatska
 Czech Republic & Slovakia / Česká republika & Slovensko
Czech Republic & Slovakia / Česká republika & Slovensko
 France / France
France / France
 Germany / Deutschland
Germany / Deutschland
 Greece / ΕΛΛΑΔΑ
Greece / ΕΛΛΑΔΑ
 Hungary / Hungary
Hungary / Hungary
 Italy / Italia
Italy / Italia
 Netherlands / Nederland
Netherlands / Nederland
 Nordic / Nordic
Nordic / Nordic
 Poland / Polska
Poland / Polska
 Portugal / Portugal
Portugal / Portugal
 Romania & Moldova / România & Moldova
Romania & Moldova / România & Moldova
 Slovenia / Slovenija
Slovenia / Slovenija
 Serbia & Montenegro / Србија и Црна Гора
Serbia & Montenegro / Србија и Црна Гора
 Spain / España
Spain / España
 Switzerland / Schweiz
Switzerland / Schweiz
 Turkey / Türkiye
Turkey / Türkiye
 UK & Ireland / UK & Ireland
UK & Ireland / UK & Ireland
 Brazil / Brasil
Brazil / Brasil
 Canada / Canada
Canada / Canada
 Latin America / Latinoamérica
Latin America / Latinoamérica
 USA / USA
USA / USA
 China / 中国
China / 中国
 India / भारत गणराज्य
India / भारत गणराज्य
 Pakistan / Pākistān
Pakistan / Pākistān
 Vietnam / Việt Nam
Vietnam / Việt Nam
 ASEAN / ASEAN
ASEAN / ASEAN
 Israel / מְדִינַת יִשְׂרָאֵל
Israel / מְדִינַת יִשְׂרָאֵל
 Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
 Middle East / Middle East
Middle East / Middle East

































To post a reply please login or register