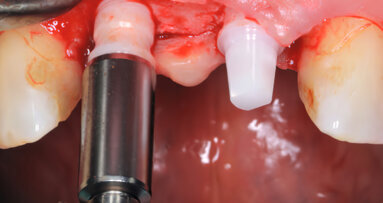
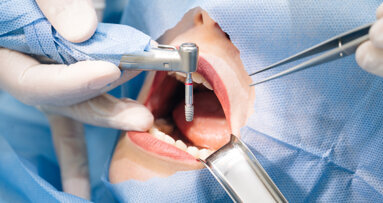
GENEVA, Switzerland: The non-profit CleanImplant Foundation will be present at the 29th Annual Scientific Meeting of the European Association for Osseointegration (EAO) and will be providing information at Booth F50, where it will continue its global awareness campaign about production-related contamination on new, sterile-packaged implants. In addition, the foundation will seek to obtain more implants to include in its current scientific study on the quality of dental implants, its fifth study overall.
In what has become the most extensive overview of surface cleanliness performance across the entire industry, the surfaces of commercially available implant systems are examined for concerning remnants from production processes. For this examination, complex, state-of-the-art scanning electron microscope analyses are conducted in collaboration with internationally renowned universities such as Charité—Universitätsmedizin Berlin in Germany.
To ensure a comprehensive overview of the manufacturing quality of the implants available on the market, the implant types to be tested are selected in advance by the foundation’s scientific advisory board. All implant manufacturers who have already provided implants for previous studies are invited to also participate in the current study, and the foundation is calling for the participation of numerous other implant manufacturers. The large-scale quality assessment study will begin when the collection process is complete.
The results of previous studies are alarming. Over a third of all sterile-packaged and ready-to-use implant types exhibited significant levels of foreign particles and residues on their surfaces.
However, there is an easy way to avoid purchasing untested, risky implants. At the CleanImplant Foundation booth at the EAO congress, experts will provide information about the impact of contaminants and advise on selecting the best implant systems. The CleanImplant Foundation creates transparency and awards its Trusted Quality Mark only to implant systems having a high-quality surface. To date, selected implant types carrying the coveted seal come from manufacturers such as Biotech Dental, bredent-medical, BTI, Camlog, Global D, medentis medical, MegaGen, NucleOSS, Sweden & Martina, Zircon Medical and Swiss Dental Solutions. The quality seal is valid for two years. Other implant systems are currently undergoing the testing process.
Tags:
VIENNA, Austria: Residues on sterile packaged implants, in particular, organic particles from the production or packaging process, are strongly suspected of...
WASHINGTON, US: The CleanImplant Foundation recently introduced its non-profit initiative at the 11th annual meeting of the International Association of ...
LONDON, UK: Introduced in 2006, the whiteSKY system from bredent has proved its efficiency in clinical practice and scientific studies. At the congress of ...
MONACO: Osstem Implant has announced its participation in the 32nd Annual Scientific Meeting of the European Association for Osseointegration (EAO 2025), ...
COMO, Italy: In November last year, 21 experts from 16 countries gathered in Como for a day of lectures, discussions and debate. The Ambassadors’ Summit, ...
KREUZLINGEN, Switzerland: For the third time this year, the CleanImplant Foundation has awarded its Trusted Quality Mark for outstanding surface ...
The CleanImplant Foundation seeks to promote global awareness of contaminated implant surfaces caused by poor quality control during production and ...
VIENNA, Austria: Under the theme of “From the past to the future”, the Osteology Foundation welcomed around 350 dental professionals at the Hofstallung ...
BERLIN, Germany: The CleanImplant Foundation, an established authority in evaluating the quality and cleanliness of implant surfaces, has awarded its ...
MONACO: The European Association for Osseointegration (EAO) will return to Monaco for its 32nd annual scientific meeting, being held from 18 to 20 ...
GLASGOW, UK: At the recent British Orthodontic Conference in Glasgow, the British Orthodontic Society (BOS) and the Oral Health Foundation (OHF) have taken...
Live webinar
Wed. 3 June 2026
1:00 pm EST (New York)
Live webinar
Thu. 4 June 2026
2:00 pm EST (New York)
Live webinar
Mon. 8 June 2026
12:00 pm EST (New York)
Live webinar
Mon. 8 June 2026
1:00 pm EST (New York)
Dr. Anthony Mak B.D.S, Prof. Marleen Peumans
Live webinar
Mon. 8 June 2026
2:00 pm EST (New York)
Live webinar
Wed. 10 June 2026
11:00 am EST (New York)
Live webinar
Wed. 10 June 2026
2:00 pm EST (New York)
Nacho Fernández-Baca DDS, MSc



 Austria / Österreich
Austria / Österreich
 Bosnia and Herzegovina / Босна и Херцеговина
Bosnia and Herzegovina / Босна и Херцеговина
 Bulgaria / България
Bulgaria / България
 Croatia / Hrvatska
Croatia / Hrvatska
 Czech Republic & Slovakia / Česká republika & Slovensko
Czech Republic & Slovakia / Česká republika & Slovensko
 France / France
France / France
 Germany / Deutschland
Germany / Deutschland
 Greece / ΕΛΛΑΔΑ
Greece / ΕΛΛΑΔΑ
 Hungary / Hungary
Hungary / Hungary
 Italy / Italia
Italy / Italia
 Netherlands / Nederland
Netherlands / Nederland
 Nordic / Nordic
Nordic / Nordic
 Poland / Polska
Poland / Polska
 Portugal / Portugal
Portugal / Portugal
 Romania & Moldova / România & Moldova
Romania & Moldova / România & Moldova
 Slovenia / Slovenija
Slovenia / Slovenija
 Serbia & Montenegro / Србија и Црна Гора
Serbia & Montenegro / Србија и Црна Гора
 Spain / España
Spain / España
 Switzerland / Schweiz
Switzerland / Schweiz
 Turkey / Türkiye
Turkey / Türkiye
 UK & Ireland / UK & Ireland
UK & Ireland / UK & Ireland
 Brazil / Brasil
Brazil / Brasil
 Canada / Canada
Canada / Canada
 Latin America / Latinoamérica
Latin America / Latinoamérica
 USA / USA
USA / USA
 China / 中国
China / 中国
 India / भारत गणराज्य
India / भारत गणराज्य
 Pakistan / Pākistān
Pakistan / Pākistān
 Vietnam / Việt Nam
Vietnam / Việt Nam
 ASEAN / ASEAN
ASEAN / ASEAN
 Israel / מְדִינַת יִשְׂרָאֵל
Israel / מְדִינַת יִשְׂרָאֵל
 Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
 Middle East / Middle East
Middle East / Middle East







































To post a reply please login or register