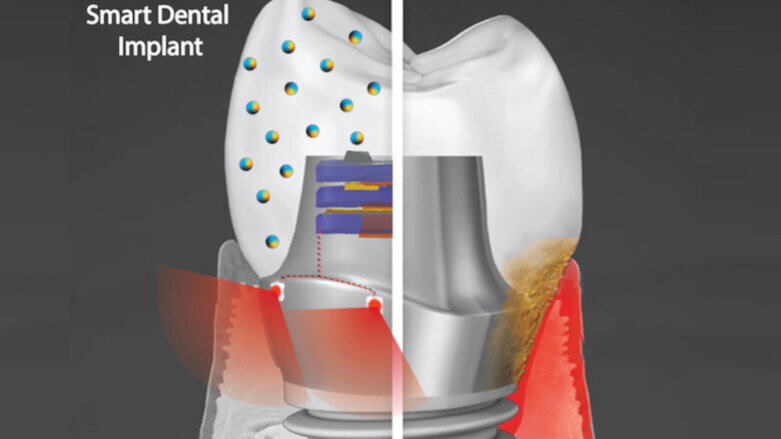
PHILADELPHIA, U.S.: In order to address implant failure and the associated effort and costs, researchers from the School of Dental Medicine of the University of Pennsylvania in Philadelphia are developing a novel smart dental implant. They are aiming for a device that resists bacterial growth and generates its own power to operate a tissue-rejuvenating light.
The new implant is designed to combine two technologies: a nanoparticle-infused material that hinders bacterial colonization and an embedded light source to conduct phototherapy, powered by natural movements of the mouth, such as when chewing or during toothbrushing. This approach is not limited to dental use, but could have other applications, such as integration into joint replacements, too.
Dr. Geelsu Hwang, assistant professor at the university and one of the researchers involved, explained in a press release: “Phototherapy can address a diverse set of health issues. But once a biomaterial is implanted, it’s not practical to replace or recharge a battery. We are using a piezoelectric material, which can generate electrical power from natural oral motions to supply a light that can conduct phototherapy, and we find that it can successfully protect gingival tissue from bacterial challenge.”
In their research, the team explored the material barium titanate (BTO), which has piezoelectric properties and is thus used in capacitators and transistors, for example. However, it has not been investigated as a basis for anti-infectious implantable biomaterials. To test its potential, the researchers embedded composite discs with nanoparticles of BTO and exposed them to Streptococcus mutans. They found that the discs hindered biofilm formation and that this effect was greater with higher concentrations of BTO.
According to the research team, BTO generates an increased negative surface charge that repels the negatively charged cell walls of bacteria and it is likely that this repulsive effect will be long-lasting. “We wanted an implant material that could resist bacterial growth for a long time because bacterial challenges are not a one-time threat,” said Dr. Hwang.
The material’s power-generating property was sustained, and when tested over long time periods, the material did not leach. It also exhibited mechanical strength comparable to that of other materials used in dentistry. In addition, the material did not harm gingival tissue, supporting the idea that it could be used without adverse effects.
In future research, the team hopes to continue to refine its smart dental implant system, test new material types and possibly use asymmetric properties on each side of the implant components in order to encourage tissue integration and resist bacterial formation. “We hope to further develop the implant system and eventually see it commercialized so it can be used in the dental field,” commented Dr. Hwang.
The research has been reported on in two studies. The most recent study, titled “Bimodal nanocomposite platform with antibiofilm and self-powering functionalities for biomedical applications,” was published in the Sept. 1, 2021, issue of ACS Applied Materials and Interfaces. The first study, titled “Human oral motion-powered smart dental implant (SDI) for in situ ambulatory photo-biomodulation therapy,” was published in the Aug. 19, 2020, issue of Advanced Healthcare Materials.
Tags:
OAK RIDGE, Tenn., U.S.: Researchers have been using neutron scattering to study how nanoparticles with antibacterial properties could be added to adhesive ...
GOTHENBURG, Sweden: Biofilm formation on dental implants is a major challenge for dental professionals. It causes patients great inconvenience and entails ...
GUILDFORD, UK: Dental disease identification is often cumbersome and time-consuming. To assist dental professionals in better detecting dental problems, ...
HANNOVER, Germany: Researchers in Germany are developing customised dental implants designed to match each patient’s individual jaw structure. The ...
DUNEDIN, New Zealand: In New Zealand, one of most common methods for treating dental caries in primary teeth is the Hall technique. The non-invasive method ...
BIRMINGHAM, UK: The link between periodontal disease and a range of systemic health conditions such as cardiovascular disease, Type 2 diabetes and ...
KAUNAS, Lithuania: The work of dental professionals was significantly affected by the COVID-19 outbreak. For this reason, researchers from Lithuania have ...
LONDON, UK: Unlike many other tissues in the human body, dental enamel does not regenerate itself once it is damaged. In what could potentially be a ...
EDMONTON, Canada: A team of researchers at the University of Alberta has secured funding to develop a 3D ultrasound device that would allow dentists to ...
URBANA, Ill., U.S.: Among the most prevalent chronic diseases in both children and adults is dental caries, which occurs when the mouth is in a state of ...
Live webinar
Tue. 26 May 2026
12:00 pm EST (New York)
Live webinar
Tue. 26 May 2026
2:00 pm EST (New York)
Live webinar
Wed. 27 May 2026
8:00 am EST (New York)
Live webinar
Thu. 28 May 2026
12:00 pm EST (New York)
Dr. Anthony Viazis DDS, MS. Inventor & Founder of Fastbraces®
Live webinar
Mon. 1 June 2026
1:00 pm EST (New York)
Live webinar
Wed. 3 June 2026
1:00 pm EST (New York)
Live webinar
Thu. 4 June 2026
2:00 pm EST (New York)



 Austria / Österreich
Austria / Österreich
 Bosnia and Herzegovina / Босна и Херцеговина
Bosnia and Herzegovina / Босна и Херцеговина
 Bulgaria / България
Bulgaria / България
 Croatia / Hrvatska
Croatia / Hrvatska
 Czech Republic & Slovakia / Česká republika & Slovensko
Czech Republic & Slovakia / Česká republika & Slovensko
 France / France
France / France
 Germany / Deutschland
Germany / Deutschland
 Greece / ΕΛΛΑΔΑ
Greece / ΕΛΛΑΔΑ
 Hungary / Hungary
Hungary / Hungary
 Italy / Italia
Italy / Italia
 Netherlands / Nederland
Netherlands / Nederland
 Nordic / Nordic
Nordic / Nordic
 Poland / Polska
Poland / Polska
 Portugal / Portugal
Portugal / Portugal
 Romania & Moldova / România & Moldova
Romania & Moldova / România & Moldova
 Slovenia / Slovenija
Slovenia / Slovenija
 Serbia & Montenegro / Србија и Црна Гора
Serbia & Montenegro / Србија и Црна Гора
 Spain / España
Spain / España
 Switzerland / Schweiz
Switzerland / Schweiz
 Turkey / Türkiye
Turkey / Türkiye
 UK & Ireland / UK & Ireland
UK & Ireland / UK & Ireland
 Brazil / Brasil
Brazil / Brasil
 Canada / Canada
Canada / Canada
 Latin America / Latinoamérica
Latin America / Latinoamérica
 USA / USA
USA / USA
 China / 中国
China / 中国
 India / भारत गणराज्य
India / भारत गणराज्य
 Pakistan / Pākistān
Pakistan / Pākistān
 Vietnam / Việt Nam
Vietnam / Việt Nam
 ASEAN / ASEAN
ASEAN / ASEAN
 Israel / מְדִינַת יִשְׂרָאֵל
Israel / מְדִינַת יִשְׂרָאֵל
 Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
 Middle East / Middle East
Middle East / Middle East



































To post a reply please login or register