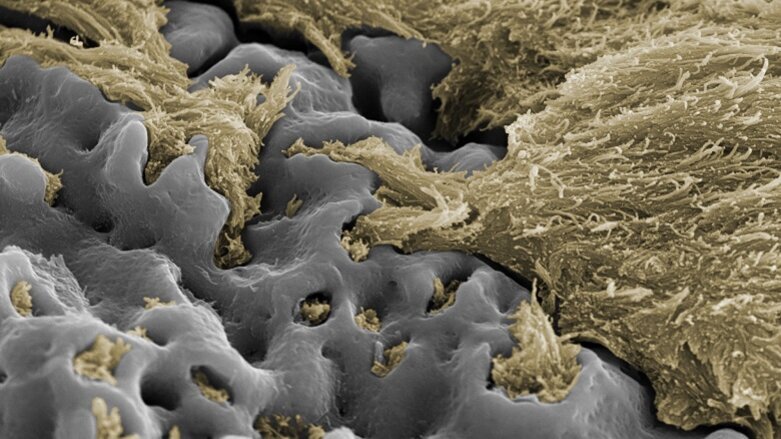
ZURICH, Switzerland: A new systematic review and meta-analysis has provided evidence confirming the efficacy of Nobel Biocare implants with the TiUnite implant surface. The study, by Prof. Matthias Karl of Saarland University in Germany and Prof. Tomas Albrektsson of the University of Gothenburg in Sweden, analysed patient results from 106 peer-reviewed publications on prospective clinical studies assessing TiUnite surface implants.
This new meta-analysis represents the largest such review of a single brand of dental implants published to date, evaluating data on 12,803 TiUnite surface implants and 4,694 patients. The results confirm that implants with the TiUnite surface have a remarkably low early failure rate and support long-term clinical survival, with early implant and patient level survival rate estimates both exceeding 99 per cent at one year, and a late implant survival rate estimate of 95.1 per cent after ten years.
Of the 106 studies, 47 evaluated biological complications. Of these 47 papers, 19 reported cases of peri-implantitis, and only in 5.2 per cent of patients. Karl and Albrektsson postulated that, assuming that any findings of peri-implantitis in the other included studies would have been reported by the authors, the actual rate of peri-implantitis among the 4,694 patients in all 106 studies would have been as low as 1.36 per cent. This is in line with an earlier report by Albrektsson of a rate of 1–2 per cent of peri-implantitis affecting well-documented implants at ten years.
Bone level change estimates of -0.4 mm at the one-year follow-up and -0.9 mm at the five-year follow-up (implant level) show that TiUnite is a surface that promotes a healthy bone response in the first year and stable bone levels long term.
“Nobel Biocare stands for high-quality products based on high-quality science. This meta-analysis unequivocally confirms what extensive internal testing and external validation have documented for over 15 years: that the TiUnite surface supports peri-implant health, bone maintenance and overall implant success long term,” said Nobel Biocare President Hans Geiselhöringer.
He added, “Such results further demonstrate the success of Nobel Biocare’s patient-centric approach to the development of products and solutions. This study serves as the strongest evidence to date that dental professionals and their patients can rely on Nobel Biocare implants with the TiUnite surface for excellent treatment outcomes.”
The study, titled “Clinical performance of dental implants with a moderately rough (TiUnite) surface: A meta-analysis of prospective clinical studies”, was published in the July/August issue of the International Journal of Oral and Maxillofacial Implants.
Tags:
MADRID, Spain: With the launch of its Xeal abutment surface and TiUltra implant surface this year, Nobel Biocare recently initiated the Mucointegration era ...
KLOTEN, Switzerland: Zygomatic implants offer a transformative solution for patients with severe maxillary bone resorption, significantly enhancing their ...
VIENNA, Austria: Nobel Biocare’s corporate forum at EuroPerio11 is set to be a highlight of the congress. Taking place on Friday, 16 May from 12:30 to ...
YORBA LINDA, Calif., U.S.: Nobel Biocare is inviting dental professionals to join what it calls the Mucointegration era with the launch of its Xeal and ...
An award-winning clinician, researcher and director at the Brånemark Centre in Perth in Australia, Dr Graham Carmichael is a consultant prosthodontist and ...
COLOGNE, Germany: Nobel Biocare has invited dental professionals to join the Mucointegration era with the launch of the Xeal and TiUltra surfaces at IDS ...
KLOTEN, Switzerland: In the ever-evolving world of dental regenerative solutions, Nobel Biocare has consistently set the benchmark for innovation and ...
KLOTEN, Switzerland: At the recent Envista Summit in Barcelona in Spain, Nobel Biocare announced its latest advancement in professional development: Zygoma ...
BREA, Calif., U.S.: Envista is proud to announce the 2020 Nobel Biocare Global Symposium, to be held in Las Vegas from April 16 to 18. Empowered by a ...
LISBON, Portugal: This year, Nobel Biocare opened a new chapter for implant dentistry with the launch of new surfaces and the introduction of the new Nobel ...
Dr. Annette Felderhoff-Fischer
Dr. Rafael Saviolo Moreira
Dr. Ian Lane Dip SED, BDS (Hons) (Ncl), MFGDP, MSc, KOIS Graduate



 Austria / Österreich
Austria / Österreich
 Bosnia and Herzegovina / Босна и Херцеговина
Bosnia and Herzegovina / Босна и Херцеговина
 Bulgaria / България
Bulgaria / България
 Croatia / Hrvatska
Croatia / Hrvatska
 Czech Republic & Slovakia / Česká republika & Slovensko
Czech Republic & Slovakia / Česká republika & Slovensko
 France / France
France / France
 Germany / Deutschland
Germany / Deutschland
 Greece / ΕΛΛΑΔΑ
Greece / ΕΛΛΑΔΑ
 Hungary / Hungary
Hungary / Hungary
 Italy / Italia
Italy / Italia
 Netherlands / Nederland
Netherlands / Nederland
 Nordic / Nordic
Nordic / Nordic
 Poland / Polska
Poland / Polska
 Portugal / Portugal
Portugal / Portugal
 Romania & Moldova / România & Moldova
Romania & Moldova / România & Moldova
 Slovenia / Slovenija
Slovenia / Slovenija
 Serbia & Montenegro / Србија и Црна Гора
Serbia & Montenegro / Србија и Црна Гора
 Spain / España
Spain / España
 Switzerland / Schweiz
Switzerland / Schweiz
 Turkey / Türkiye
Turkey / Türkiye
 UK & Ireland / UK & Ireland
UK & Ireland / UK & Ireland
 Brazil / Brasil
Brazil / Brasil
 Canada / Canada
Canada / Canada
 Latin America / Latinoamérica
Latin America / Latinoamérica
 USA / USA
USA / USA
 China / 中国
China / 中国
 India / भारत गणराज्य
India / भारत गणराज्य
 Pakistan / Pākistān
Pakistan / Pākistān
 Vietnam / Việt Nam
Vietnam / Việt Nam
 ASEAN / ASEAN
ASEAN / ASEAN
 Israel / מְדִינַת יִשְׂרָאֵל
Israel / מְדִינַת יִשְׂרָאֵל
 Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
 Middle East / Middle East
Middle East / Middle East








































To post a reply please login or register