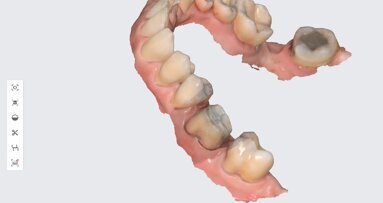
LOS ANGELES, US: As 3D printing in dentistry evolves to become industry standard, the need for access to more materials is critical. In a step that 3D-printing company SprintRay calls a “key milestone”, the US Food and Drug Administration has granted the company clearance to market its NightGuard Flex 3D-printing resin as a Class II medical device in the US.
Used to fabricate appliances such as mouth guards, night guards and splints, the resin joins SprintRay’s growing number of end-to-end dental 3D-printing solutions. Designed to provide the best possible patient experience with high-impact strength and wear-resistance, NightGuard Flex also allows same-day patient delivery, eliminates the need for third-party resins and creates uniform, end-to-end dental 3D-printing workflows.
According to SprintRay, the resin was developed, in part, to help meet the increasing demand for night guards. The COVID-19 pandemic has induced extra stress and worry, and a March 2021 survey conducted by the American Dental Association indicated that more than 70% of dentists were seeing a rise in the number of patients experiencing bruxism. In a press release, SprintRay CEO Dr Amir Mansouri said, “SprintRay’s NightGuard Flex resin has totally changed the patient and doctor experience in the treatment of bruxism and teeth grinding.”
The pandemic is still having an impact on the global community, and innovation within dentistry will be vital in ensuring that the needs of patients can be met with few difficulties and at competitive prices. This recent announcement from SprintRay comes after the company demonstrated its commitment to supporting occlusal health through its Protect a Smile campaign and after its North American distribution partnership agreement with Nobel Biocare, which as reported by Dental Tribune International, will result in reduced implant delivery times.
“Our patient-centred design is durable, flexible, more comfortable, and enables faster delivery and greater access for patients to receive their night guards same-day, through chairside SprintRay 3D printing and at more affordable prices,” noted Dr Mansouri.
LOS ANGELES, US/COLOGNE, Germany: SprintRay is a digital dentistry company that offers chairside 3D-printing solutions, including for fabrication of crowns ...
In today’s busy life, patients may have trouble coordinating their work and home schedules when treatment requires multiple appointments. Typically, when ...
3D printing has revolutionised modern dentistry, offering a cost-effective, versatile and efficient alternative to traditional fabrication methods. However,...
LOS ANGELES, U.S.: Technological advancements in dentistry are like a motor force that drives innovation and growth. This is true for digital dentistry. ...
LOS ANGELES, US: SprintRay has announced the launch of the Pro S dental 3D printer product line in combination with RayWare Cloud and new SprintRay resins....
LOS ANGELES, U.S.: SprintRay, a 3D printer manufacturer that focuses on digital dentistry, announced last week that it has commenced a partnership with the ...
3D printing is here to stay, and it continues to be one of the major topics in dentistry. Just recently, Dental Tribune International had the pleasure of ...
Patrick Thurm is the managing director and general manager for Europe at SprintRay, a young 3D printing company that opened its European branch office in ...
MIAMI, US: At its 3DNext summit in Miami, attended by over 250 dental professionals, SprintRay, a leader in dental 3D printing, unveiled its most ...
Dentistry has undergone a remarkable transformation with the advent of 3D-printing technology. Gone are the days when patients had to endure multiple visits...
Live webinar
Fri. 19 June 2026
9:00 am EST (New York)
Theodoros Gonidis, Meni Chatzinikolaou



 Austria / Österreich
Austria / Österreich
 Bosnia and Herzegovina / Босна и Херцеговина
Bosnia and Herzegovina / Босна и Херцеговина
 Bulgaria / България
Bulgaria / България
 Croatia / Hrvatska
Croatia / Hrvatska
 Czech Republic & Slovakia / Česká republika & Slovensko
Czech Republic & Slovakia / Česká republika & Slovensko
 France / France
France / France
 Germany / Deutschland
Germany / Deutschland
 Greece / ΕΛΛΑΔΑ
Greece / ΕΛΛΑΔΑ
 Hungary / Hungary
Hungary / Hungary
 Italy / Italia
Italy / Italia
 Netherlands / Nederland
Netherlands / Nederland
 Nordic / Nordic
Nordic / Nordic
 Poland / Polska
Poland / Polska
 Portugal / Portugal
Portugal / Portugal
 Romania & Moldova / România & Moldova
Romania & Moldova / România & Moldova
 Slovenia / Slovenija
Slovenia / Slovenija
 Serbia & Montenegro / Србија и Црна Гора
Serbia & Montenegro / Србија и Црна Гора
 Spain / España
Spain / España
 Switzerland / Schweiz
Switzerland / Schweiz
 Turkey / Türkiye
Turkey / Türkiye
 UK & Ireland / UK & Ireland
UK & Ireland / UK & Ireland
 Brazil / Brasil
Brazil / Brasil
 Canada / Canada
Canada / Canada
 Latin America / Latinoamérica
Latin America / Latinoamérica
 USA / USA
USA / USA
 China / 中国
China / 中国
 India / भारत गणराज्य
India / भारत गणराज्य
 Pakistan / Pākistān
Pakistan / Pākistān
 Vietnam / Việt Nam
Vietnam / Việt Nam
 ASEAN / ASEAN
ASEAN / ASEAN
 Israel / מְדִינַת יִשְׂרָאֵל
Israel / מְדִינַת יִשְׂרָאֵל
 Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
 Middle East / Middle East
Middle East / Middle East








































To post a reply please login or register