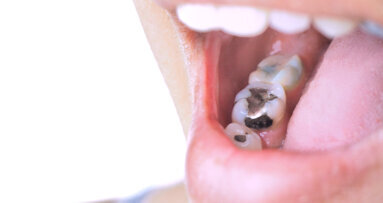
WHITE OAK, Md., U.S.: The U.S. Food and Drug Administration (FDA) has updated its guidance for the use of dental amalgam in tooth restorations. The agency said in September that the use of dental amalgam should be avoided whenever possible in certain groups, such as in pregnant women, women who are planning to become pregnant, in children and in people with certain preexisting neurological diseases.
The use of dental amalgam has remained widespread in the U.S., despite its use being phased out or banned in certain groups in a number of countries. Until its recent announcement, the FDA had considered dental amalgam restorations to be safe for adults and children over the age of 6.
As detailed in its September statement, the FDA’s new position on the use of dental amalgam is that its use may pose a greater risk for potential harmful health effects for certain groups, and it therefore recommends that its use in these groups be avoided “whenever possible and appropriate”.
These groups are:
-
pregnant women and their developing fetuses;
-
women who are planning to become pregnant;
-
nursing women and their newborns and infants;
-
children, especially those younger than 6 years of age;
-
people with preexisting neurological disease such as multiple sclerosis, Alzheimer’s disease and Parkinson’s disease;
-
people with impaired kidney function; and
-
people with known heightened sensitivity (allergy) to mercury or other components of dental amalgam.
As pointed out by Multiple Sclerosis News Today, the agency has not banned the use of dental amalgam, or explicitly stated that it should not be used, but has updated its guidance and published a new information brochure on its use and removal in tooth restorations. The FDA does not recommend the removal or replacement of dental amalgam restorations that are in good condition.
The agency’s brochure states that “studies on people with dental amalgam do not show conclusive evidence that dental amalgam causes harmful health effects in the general population.”
It has been argued within the international medical and scientific community that there is evidence of a causal link between the use of dental amalgam in tooth restorations and the incidence of neurological conditions such as multiple sclerosis. The International Academy of Oral Medicine & Toxicology has compiled a summary of research related to mercury exposure and the incidence of multiple sclerosis, and this can be accessed together with additional resources on the academy’s website.
Dental Tribune International reported in May that 9,287 pounds (4,212 kg) of mercury was used for dental amalgam in the U.S. in 2018. According to the U.S. Environmental Protection Agency, this accounted for 46.8% of the total elemental mercury that was used to make products in the country in that year.
Tags:
CHICAGO, U.S.: The American Dental Association (ADA) in December adopted an interim policy that calls for a total ban on all vaping products that are not ...
GENEVA, Switzerland: The second meeting of the Conference of the Parties (COP2) to the Minamata Convention on Mercury was held at the International ...
SAN FRANCISCO, U.S.: Dentists often refer to encouraging positive behavioral change in their patients as one of the greatest challenges they face, and GSK ...
BELFAST, UK: The phase-out of dental amalgam is currently taking place in a number of countries owing, in part, to its deleterious effects on the natural ...
SAN JUAN, Puerto Rico/SPRING, Texas, U.S.: Mercury in dental amalgam is a hidden source of global mercury pollution, resulting from factors such as the ...
STRASBOURG, France: Amalgam is the restorative material of choice for many dentists. It is the gold standard of dental care. However, its popularity has ...
MANILA, Philippines: The safety of the use of amalgam in the mouth is one of the most controversial topics in modern dentistry, and the widely ratified ...
LONDON, England: National Health Service (NHS) dental charges in England have increased once more, under amended regulations that came into force this ...
CHICAGO, US: The American Dental Association (ADA) has issued updated recommendations on dental imaging, reinforcing that radiographs and CBCT scans should ...
DRESDEN, Germany: The temperature is currently climbing to 38 °C throughout Europe. While this heatwave is gruelling, it might also influence the ...
Live webinar
Wed. 3 June 2026
1:00 pm EST (New York)
Live webinar
Thu. 4 June 2026
2:00 pm EST (New York)
Live webinar
Mon. 8 June 2026
12:00 pm EST (New York)
Live webinar
Mon. 8 June 2026
1:00 pm EST (New York)
Dr. Anthony Mak B.D.S, Prof. Marleen Peumans
Live webinar
Mon. 8 June 2026
2:00 pm EST (New York)
Live webinar
Wed. 10 June 2026
11:00 am EST (New York)
Live webinar
Wed. 10 June 2026
2:00 pm EST (New York)
Nacho Fernández-Baca DDS, MSc



 Austria / Österreich
Austria / Österreich
 Bosnia and Herzegovina / Босна и Херцеговина
Bosnia and Herzegovina / Босна и Херцеговина
 Bulgaria / България
Bulgaria / България
 Croatia / Hrvatska
Croatia / Hrvatska
 Czech Republic & Slovakia / Česká republika & Slovensko
Czech Republic & Slovakia / Česká republika & Slovensko
 France / France
France / France
 Germany / Deutschland
Germany / Deutschland
 Greece / ΕΛΛΑΔΑ
Greece / ΕΛΛΑΔΑ
 Hungary / Hungary
Hungary / Hungary
 Italy / Italia
Italy / Italia
 Netherlands / Nederland
Netherlands / Nederland
 Nordic / Nordic
Nordic / Nordic
 Poland / Polska
Poland / Polska
 Portugal / Portugal
Portugal / Portugal
 Romania & Moldova / România & Moldova
Romania & Moldova / România & Moldova
 Slovenia / Slovenija
Slovenia / Slovenija
 Serbia & Montenegro / Србија и Црна Гора
Serbia & Montenegro / Србија и Црна Гора
 Spain / España
Spain / España
 Switzerland / Schweiz
Switzerland / Schweiz
 Turkey / Türkiye
Turkey / Türkiye
 UK & Ireland / UK & Ireland
UK & Ireland / UK & Ireland
 Brazil / Brasil
Brazil / Brasil
 Canada / Canada
Canada / Canada
 Latin America / Latinoamérica
Latin America / Latinoamérica
 USA / USA
USA / USA
 China / 中国
China / 中国
 India / भारत गणराज्य
India / भारत गणराज्य
 Pakistan / Pākistān
Pakistan / Pākistān
 Vietnam / Việt Nam
Vietnam / Việt Nam
 ASEAN / ASEAN
ASEAN / ASEAN
 Israel / מְדִינַת יִשְׂרָאֵל
Israel / מְדִינַת יִשְׂרָאֵל
 Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
 Middle East / Middle East
Middle East / Middle East





















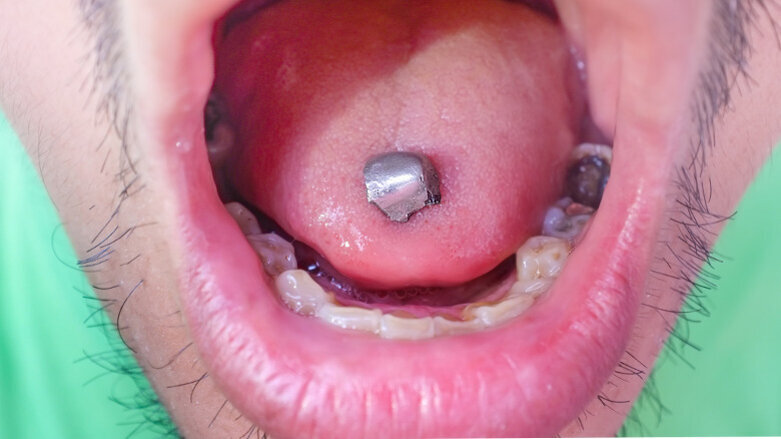





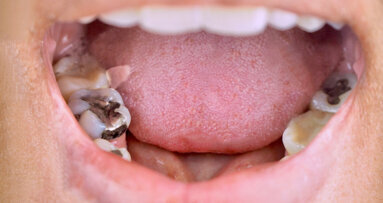
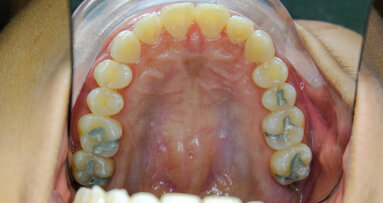







Seems odd only safe place for amalgam is in certain people otherwise hazardous waste
You would think there would be a large number of dentists with neuro issues with all spilled mercury and handling of it by “wet fingered” dentists from days of old.I have not heard of any finding.
Could someone tell me if they have read anything like that.
Will this change the dental insurance companies’ determination of payment for a composite filling?