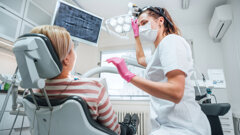
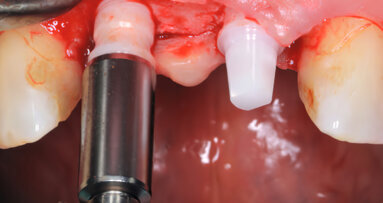
BERLN, Germany: The CleanImplant Foundation has awarded the Kontact S implant system from French manufacturer Biotech Dental with the Trusted Quality Mark. This quality seal, which confirms first-class surface purity of dental implants, is only awarded by the scientific advisory board of the CleanImplant Foundation after a rigorous analysis and testing process. The independent non-profit organisation aims to increase transparency with regard to the surface cleanliness of dental implants in the market.
Phillipe Veran, CEO and founder of Biotech Dental, was delighted with this recognition: “At Biotech Dental, we have always been committed to the highest quality standards in implant design and production processes. We will never accept compromise in our product quality. We owe it to our customers and patients.”
A growing number of companies and dentists support the CleanImplant Foundation. The results of the major quality assessments conducted by Dr Dirk U. Duddeck, founder and head of research at CleanImplant, show that a clean implant surface cannot be taken for granted. Unfortunately, there are still far too many sterile-packaged implant systems of inferior quality with considerable impurities from the production and or packaging process, according to Dr Duddeck. “If implant manufacturers claim the highest quality standards, they must also guarantee these in terms of the cleanliness of implant surfaces. Only then, can manufacturers live up to their quality promise to practitioners and patients 100%,” he said.
To date, a total of 12 implant systems from well-known manufacturers have received the Trusted Quality seal for their products after implementing the CleanImplant consensus-based guideline in its production processes.
The certification has been awarded for selected implant systems made by the following manufacturers: bredent medical, BTI, Camlog, Global D, medentis medical, MegaGen, Nobel Biocare, NucleOSS, Sweden & Martina, Straumann, Zircon Medical and SDS. Most recently, the CeramTec Group was awarded the newly established Certified Production Quality seal—a mark that recognises outstanding production standards in manufacturing.
KREUZLINGEN, Switzerland: For the third time this year, the CleanImplant Foundation has awarded its Trusted Quality Mark for outstanding surface ...
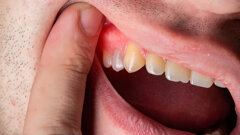
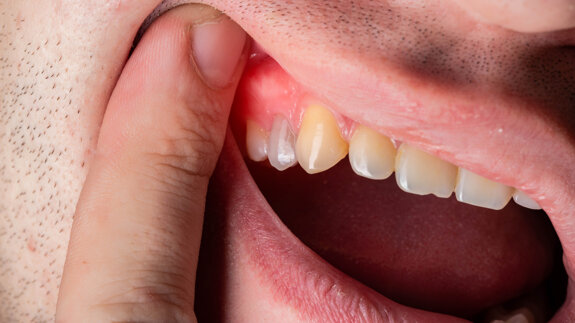
VIENNA, Austria: Residues on sterile packaged implants, in particular, organic particles from the production or packaging process, are strongly suspected of...
LONDON, UK: Introduced in 2006, the whiteSKY system from bredent has proved its efficiency in clinical practice and scientific studies. At the congress of ...
HÜNENBERG, Switzerland: TRI Dental Implants recently announced that it has obtained CE mark certification, enabling it to market its digital implant, ...
FORT LAUDERDALE, Fla., US: Swiss dental implant specialist Patent Medical has been named “Dental Implants Manufacturer of the Year in Europe” by MedTech...
PRAGUE, Czech Republic: Osstem Europe, the European headquarters of Osstem Implant, one of the world’s major dental implant manufacturers, has introduced ...
SALON-DE-PROVENCE, France : Monday, 19 June, was a beautiful sunny day in Salon-de-Provence, when orthodontists, collaborators, partners, entrepreneurs and ...
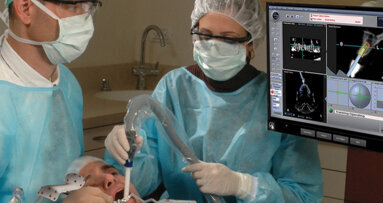
NEW YORK, U.S.: Image Navigation has announced a new image-guided implant (IGI) dentistry system that extends the use of CBCT scanners to include ...
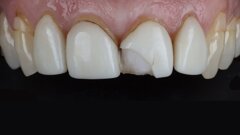
Prior to marketing the CLEARFIL MAJESTY ES Flow series, Kuraray Noritake Dental had to prove that the material meets the standards of ISO 4049:2019 for ...
COLOGNE, Germany: Vista Dental Products, a US-based manufacturer, is seeking international distribution partners. Since entering into business in 1997, ...
Live webinar
Thu. 2 April 2026
12:00 pm EST (New York)
Live webinar
Wed. 8 April 2026
1:00 pm EST (New York)
Live webinar
Thu. 9 April 2026
1:00 pm EST (New York)
Live webinar
Thu. 9 April 2026
2:00 pm EST (New York)
Prof. Moritz Kebschull, Cat Edney
Live webinar
Fri. 10 April 2026
10:00 am EST (New York)
Live webinar
Fri. 10 April 2026
11:00 am EST (New York)
Dr. med. dent. Henrik-Christian Carl Hollay
Live webinar
Fri. 10 April 2026
12:00 pm EST (New York)
Prof. Dr. Ali Murat Kökat



 Austria / Österreich
Austria / Österreich
 Bosnia and Herzegovina / Босна и Херцеговина
Bosnia and Herzegovina / Босна и Херцеговина
 Bulgaria / България
Bulgaria / България
 Croatia / Hrvatska
Croatia / Hrvatska
 Czech Republic & Slovakia / Česká republika & Slovensko
Czech Republic & Slovakia / Česká republika & Slovensko
 France / France
France / France
 Germany / Deutschland
Germany / Deutschland
 Greece / ΕΛΛΑΔΑ
Greece / ΕΛΛΑΔΑ
 Hungary / Hungary
Hungary / Hungary
 Italy / Italia
Italy / Italia
 Netherlands / Nederland
Netherlands / Nederland
 Nordic / Nordic
Nordic / Nordic
 Poland / Polska
Poland / Polska
 Portugal / Portugal
Portugal / Portugal
 Romania & Moldova / România & Moldova
Romania & Moldova / România & Moldova
 Slovenia / Slovenija
Slovenia / Slovenija
 Serbia & Montenegro / Србија и Црна Гора
Serbia & Montenegro / Србија и Црна Гора
 Spain / España
Spain / España
 Switzerland / Schweiz
Switzerland / Schweiz
 Turkey / Türkiye
Turkey / Türkiye
 UK & Ireland / UK & Ireland
UK & Ireland / UK & Ireland
 Brazil / Brasil
Brazil / Brasil
 Canada / Canada
Canada / Canada
 Latin America / Latinoamérica
Latin America / Latinoamérica
 USA / USA
USA / USA
 China / 中国
China / 中国
 India / भारत गणराज्य
India / भारत गणराज्य
 Pakistan / Pākistān
Pakistan / Pākistān
 Vietnam / Việt Nam
Vietnam / Việt Nam
 ASEAN / ASEAN
ASEAN / ASEAN
 Israel / מְדִינַת יִשְׂרָאֵל
Israel / מְדִינַת יִשְׂרָאֵל
 Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
 Middle East / Middle East
Middle East / Middle East





























To post a reply please login or register