The oral microbiome is an incredibly complex bacterial community. In total, it harbours over 700 bacterial taxa.[i] The oral microbiome with its various microorganisms exists in the form of a biofilm, that is, microbial structures readily forming on all hard- and non-shedding surfaces in a fluid system. For this reason, the oral cavity with its many surfaces, including the hard tissue of teeth, hard substance of implants and crowns, and soft tissue of the oral mucosa and the tongue, represents the ideal ground for microbes to colonise and biofilm to develop.
This development is mediated through the formation of a salivary pellicle, followed by subsequent colonisation of single bacteria, which proliferate, agglutinate and form complexes in which an intense exchange of molecules occurs. In a state of equilibrium, oral biofilm plays a crucial role in maintaining oral homeostasis and preventing disease development. However, with time, biofilm provides its own metabolic products to attract secondary plaque bacteria with high pathogenic potential that can ultimately lead to certain imbalances. Once the complex biofilm, the maturation of which may take weeks or even months, fully exercises its pathogenic potential, the oral microbiome can be disrupted, which in turn leads to dysbiosis and causes disease in the mouth and elsewhere in the body.
Role of oral biofilm in the aetiology of disease
Oral biofilm is medically important because it has broad implications for maintaining not only oral health but also systemic health. It is estimated that biofilms account for over 80% of microbial infections in the body.[ii] Research has undoubtedly shown that biofilm-dependent infections, including oral diseases such as caries, and periodontal and endodontic disease, thus pose major public health and cost concerns globally.
“There’s only one way to address this problem: research, research and research.”
Periodontal disease, in particular, is considered one of the most prevalent diseases worldwide, affecting up to 50% of the world’s adult population, according to FDI World Dental Federation. Previous research has established connections between periodontal disease and preterm births, low birthweight, diabetes, and risk factors associated with cardiovascular disease and stroke. Globally, periodontitis is responsible for an estimated US$54 billion per year in lost productivity and a major portion of the US$442 billion spent annually on oral disease, according to the European Federation of Periodontology.
Despite the success in prevention of oral disease over the past decades, the prevalence of periodontal disease remains high. In an interview with Dental Tribune International, Prof. Thomas Beikler, Director of the Department of Periodontics, Preventive and Restorative Dentistry at the University Medical Center Hamburg-Eppendorf in Germany, said: “The enormous efforts in the past to promote oral hygiene around the globe have fortunately resulted in a drastic reduction of caries prevalence; however, periodontal disease seems not to have been affected to the same extent. Therefore, there’s only one way to address this problem: research, research and research.”
Control of oral biofilm
Owing to the vast implications of oral biofilm for dental and general health, its effective control represents a major challenge. There is no question that the formation and maturation of oral biofilm need to be tackled by dental professionals across all specialties in all countries worldwide.
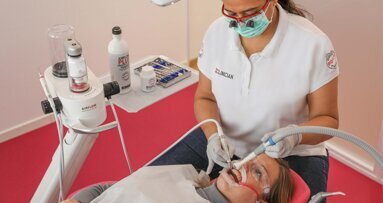
“The backbone of any therapeutic and preventive approach is the disruption of the intra-oral biofilm. An example of technologies that have been successfully demonstrated to aid supra- and subgingival instrumentation are air polishing devices with low-abrasive powders that can disrupt the biofilm on all intra-oral surfaces, including mucous membranes and difficult-to-reach subgingival areas, without harming the hard tissue and the even more vulnerable soft tissue,” Beikler explained. “Their effects can be augmented by antimicrobial and immune- and microbiome-modulating strategies. Regarding the latter, probiotics offer an interesting and promising alternative to the use of unspecific antimicrobial agents like chlorhexidine or phenolic compounds. However, additional research, that is, appropriately designed and well-structured multicentre clinical trials, is critically needed to fully appraise their therapeutic effects,” he continued.
Another major challenge that needs to be taken into consideration in this respect is that trying to eliminate a specific pathogen using antimicrobial agents may disturb the sensitive microbial ecology and eventually induce microbial dysbiosis of the oral cavity.
“At this point in time, no specific therapy is known to reverse an intra-oral dysbiosis and to successfully re-establish a eubiotic oral microbiota. Although many hypotheses exist, we still don’t know the exact mechanisms that cause an intra-oral dysbiosis and, as a consequence, unfortunately don’t have a specific strategy to reverse it. However, there is no doubt that home and professional biofilm management, that is, the regular quantitative suppression of biofilm formation, are beneficial to the patient and a hallmark of preventive dentistry,” said Beikler.
Education on oral biofilm
Until the early 1990s, dentists confined their treatment for combating oral plaque to removing calculus twice a year because there was only limited understanding of biofilm. Over the past decades, research on and improved knowledge about the parameters that determine the development of oral disease have spurred the development of new preventive strategies. For instance, piezoceramic instruments and low-abrasion powders have made professional biofilm management safer, minimally invasive and more comfortable for both the patient and the dental practitioner. Moreover, the launch and further development of AIRFLOW devices by EMS fundamentally changed biofilm management and eventually resulted in the development of Guided Biofilm Therapy, an innovative concept for contemporary prophylaxis.
“It is clear that all dental disciplines, ranging from dental hygienists to orthodontists, are highly affected by oral biofilm.”
As research and development of new technologies and therapies continues to progress, so too does knowledge in the dental professional community need to advance for effective integration of these developments into everyday practice for the well-being of the patient.
“As biofilms can be found on all oral surfaces, it is clear that all dental disciplines, ranging from dental hygienists to orthodontists, are highly affected by oral biofilm and its sequelae,” highlighted Beikler. “Unfortunately, prevention is sometimes still considered to be toothbrushing augmented by some kind of professional tooth cleaning. However, the whole oral cavity should be the therapeutic focus of a systematic preventive approach.”
In order to educate dental professionals worldwide, the Swiss Dental Academy will broadcast the first 24-hour webinar in dentistry shortly. Starting on 23 November at 1 p.m. CET, over 30 expert speakers will be lecturing online about the latest in preventive dentistry for one whole day. Under the title “Modulation of intra-oral biofilm”, Beikler will be giving a presentation on 23 November at 5 p.m. CET. “This lecture aims to enhance awareness of the fact that the quantitative reduction of biofilm around teeth by toothbrushes and some sort of professional tooth cleaning is definitely not sufficient and that systematic approaches are needed to reduce the prevalence of oral disease. Moreover, I will be giving a short overview of strategies that have the potential to modulate the oral microbiome.”
[i] Latest figures available (www.homd.org).
[ii] According to the US National Institutes of Health (www.grants.nih.gov).
Tags:



 Austria / Österreich
Austria / Österreich
 Bosnia and Herzegovina / Босна и Херцеговина
Bosnia and Herzegovina / Босна и Херцеговина
 Bulgaria / България
Bulgaria / България
 Croatia / Hrvatska
Croatia / Hrvatska
 Czech Republic & Slovakia / Česká republika & Slovensko
Czech Republic & Slovakia / Česká republika & Slovensko
 France / France
France / France
 Germany / Deutschland
Germany / Deutschland
 Greece / ΕΛΛΑΔΑ
Greece / ΕΛΛΑΔΑ
 Hungary / Hungary
Hungary / Hungary
 Italy / Italia
Italy / Italia
 Netherlands / Nederland
Netherlands / Nederland
 Nordic / Nordic
Nordic / Nordic
 Poland / Polska
Poland / Polska
 Portugal / Portugal
Portugal / Portugal
 Romania & Moldova / România & Moldova
Romania & Moldova / România & Moldova
 Slovenia / Slovenija
Slovenia / Slovenija
 Serbia & Montenegro / Србија и Црна Гора
Serbia & Montenegro / Србија и Црна Гора
 Spain / España
Spain / España
 Switzerland / Schweiz
Switzerland / Schweiz
 Turkey / Türkiye
Turkey / Türkiye
 UK & Ireland / UK & Ireland
UK & Ireland / UK & Ireland
 Brazil / Brasil
Brazil / Brasil
 Canada / Canada
Canada / Canada
 Latin America / Latinoamérica
Latin America / Latinoamérica
 USA / USA
USA / USA
 China / 中国
China / 中国
 India / भारत गणराज्य
India / भारत गणराज्य
 Pakistan / Pākistān
Pakistan / Pākistān
 Vietnam / Việt Nam
Vietnam / Việt Nam
 ASEAN / ASEAN
ASEAN / ASEAN
 Israel / מְדִינַת יִשְׂרָאֵל
Israel / מְדִינַת יִשְׂרָאֵל
 Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
 Middle East / Middle East
Middle East / Middle East














































To post a reply please login or register