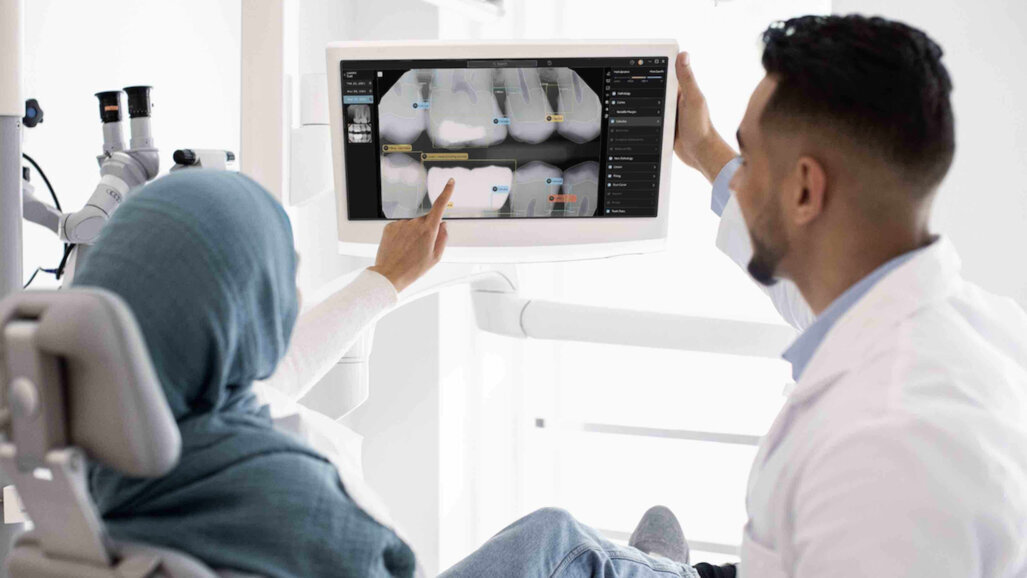
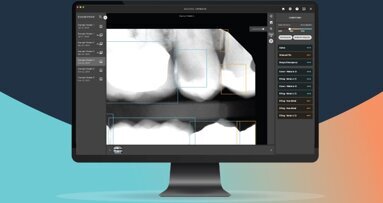
LOS ANGELES, US/DUBAI, UAE: The UAE Ministry of Health and Prevention has authorised the use of Second Opinion, an artificial intelligence (AI) tool that assists in dental radiology. Produced by the company Pearl, the device is designed to help dental professionals scan and detect a wide range of dental conditions using radiographs.
Second Opinion applies computer vision technologies to detect a comprehensive array of pathologies, existing restorations and natural anatomy in dental radiographs. According to the company, the system analyses radiographs in real time, allowing dentists to display, review, add or reject the system’s detections while conducting examinations.
“With Second Opinion cleared, patients in the UAE can begin to gain the many benefits of AI-assisted dentistry and greater assurance in quality of care at Second Opinion-equipped dental practices,” said Dr Oussama Alaoui, head of prosthodontics at Harmony Medical Center in Abu Dhabi, in a press release. He added: “I look forward to witnessing the revolutionary impact and advantages that AI will bring to our dental care.”
Ophir Tanz, CEO and founder of Pearl, commented on the market launch: “Our Second Opinion technology enables more efficient and transparent doctor–patient communication while facilitating a higher standard of care. With the implementation of the platform in the UAE, we are excited to take a first step towards driving growth and better patient outcomes across Middle Eastern dentistry.”
In an interview, Tanz said that Second Opinion improves doctor–patient communication as dentists can clearly communicate their diagnosis with the support of the tool, giving their patients a greater sense of assurance. “It eliminates the doubt that makes patients seek a second opinion elsewhere—that is why we call it Second Opinion,” he explained.
The latest clearance follows the solution’s CE-marking in Europe, its Medical Device Establishment License for use in Canada, its clearance from the Therapeutic Goods Administration in Australia and, in New Zealand, its clearance from the New Zealand Medicines and Medical Devices Safety Authority.
More information on Second Opinion can be found at www.hellopearl.com/products/second-opinion.
Tags:
LOS ANGELES, US: Health regulators in New Zealand and Australia have provided market authorisation for Second Opinion, an artificial intelligence (AI) tool ...
BOSTON, US: Overjet, a dental start-up that develops software powered by artificial intelligence (AI), recently announced that it has received 510(k) ...
COLOGNE, Germany: Pearl, a leader in artificial intelligence (AI) systems for dental practices, announced today at IDS 2021 that its Second Opinion AI ...
CHARLOTTE, N.C., US: Dentsply Sirona, the world’s largest diversified manufacturer of dental products and solutions, and Pearl, a leader in dental ...
CHARLOTTE, N.C., US: Dentsply Sirona and Siemens Healthineers have announced that their dental MRI system, MAGNETOM Free.Max Dental Edition, has received ...
WEST HOLLYWOOD, Calif., US: Second Opinion, an artificial intelligence (AI) diagnostic tool that assists in dental radiography, has been cleared by the US ...
ZURICH, Switzerland: Swiss-based start-up Odne has recently announced that it has received market clearance from the US Food and Drug Administration (FDA) ...
LOS ANGELES, US: As 3D printing in dentistry evolves to become industry standard, the need for access to more materials is critical. In a step that ...
KIEL, Germany: In November last year, researchers at Kiel University reported a technological advance in additive manufacturing. They reported the first ...
LOS ANGELES, US: Pearl, a global leader in dental artificial intelligence (AI) solutions, recently announced that it has raised US$58 million (€53.4 ...
Live webinar
Wed. 3 June 2026
1:00 pm EST (New York)
Live webinar
Thu. 4 June 2026
2:00 pm EST (New York)
Live webinar
Mon. 8 June 2026
12:00 pm EST (New York)
Live webinar
Mon. 8 June 2026
1:00 pm EST (New York)
Dr. Anthony Mak B.D.S, Prof. Marleen Peumans
Live webinar
Mon. 8 June 2026
2:00 pm EST (New York)
Live webinar
Wed. 10 June 2026
11:00 am EST (New York)
Live webinar
Wed. 10 June 2026
2:00 pm EST (New York)
Nacho Fernández-Baca DDS, MSc



 Austria / Österreich
Austria / Österreich
 Bosnia and Herzegovina / Босна и Херцеговина
Bosnia and Herzegovina / Босна и Херцеговина
 Bulgaria / България
Bulgaria / България
 Croatia / Hrvatska
Croatia / Hrvatska
 Czech Republic & Slovakia / Česká republika & Slovensko
Czech Republic & Slovakia / Česká republika & Slovensko
 France / France
France / France
 Germany / Deutschland
Germany / Deutschland
 Greece / ΕΛΛΑΔΑ
Greece / ΕΛΛΑΔΑ
 Hungary / Hungary
Hungary / Hungary
 Italy / Italia
Italy / Italia
 Netherlands / Nederland
Netherlands / Nederland
 Nordic / Nordic
Nordic / Nordic
 Poland / Polska
Poland / Polska
 Portugal / Portugal
Portugal / Portugal
 Romania & Moldova / România & Moldova
Romania & Moldova / România & Moldova
 Slovenia / Slovenija
Slovenia / Slovenija
 Serbia & Montenegro / Србија и Црна Гора
Serbia & Montenegro / Србија и Црна Гора
 Spain / España
Spain / España
 Switzerland / Schweiz
Switzerland / Schweiz
 Turkey / Türkiye
Turkey / Türkiye
 UK & Ireland / UK & Ireland
UK & Ireland / UK & Ireland
 Brazil / Brasil
Brazil / Brasil
 Canada / Canada
Canada / Canada
 Latin America / Latinoamérica
Latin America / Latinoamérica
 USA / USA
USA / USA
 China / 中国
China / 中国
 India / भारत गणराज्य
India / भारत गणराज्य
 Pakistan / Pākistān
Pakistan / Pākistān
 Vietnam / Việt Nam
Vietnam / Việt Nam
 ASEAN / ASEAN
ASEAN / ASEAN
 Israel / מְדִינַת יִשְׂרָאֵל
Israel / מְדִינַת יִשְׂרָאֵל
 Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
 Middle East / Middle East
Middle East / Middle East

































To post a reply please login or register