Mineral trioxide aggregate (MTA) was introduced as an alternative to traditional materials for the repair of root perforations and pulp capping and as a retrograde root filling owing to its superior biocompatibility and ability to seal the root canal system. Traditionally, calcium hydroxide (Ca(OH)2) has been the material of choice for the apexification of immature permanent teeth but MTA holds significant promise as an alternative to multiple treatments with Ca(OH)2. This paper discusses the use of Ca(OH)2 as a traditional apexification material and provides an overview of the composition, properties and applications of MTA with emphasis on its use in the apexification of immature permanent teeth. A case report is presented to demonstrate its use.
Introduction
Trauma causes cessation of root development and fragile root canals become weak, making it difficult to create an artificial barrier or induce closure of apical foramina with calcified tissue. [1] MTA was first described in dental scientific literature in 1993 [2] and was given approval for endodontic use by the US Food and Drug Administration in 1998. Up to 2002, only one MTA material, consisting of grey coloured powder, was available and then white MTA was introduced. Both formulae contain 75% Portland cement, 20% bismuth oxide and 5% gypsum by weight.
The aim of apexification is the production of mineralized apical tissue and to limit bacterial infection in immature anterior tooth. The inadequacy of Ca(OH)2 apexification owing to the need for multiple visits for refreshment and reinfection because of its temporary seal [3,4] led to the use of MTA, which forms a barrier and prevents microleakage. It is biocompatible and facilitates the formation of dentinal bridges and cementum, and regeneration of the periodontal ligament. [5] It has the ability to stimulate cytokine release from the bone cells, indicating that it actively promotes hard tissue formation. [6]
embedImagecenter("Imagecenter_1_1317",1317, "large");
Case report
A 14-year-old female patient suffering from painful symptoms caused by her maxillary central incisorswas examined in the Department of Pediatic Dentistry and Orthodontics of Al-Quds University in Jerusalem for evaluation and treatment.
Investigation revealed a trauma four years before associated with an enamel–dentine fracture. No treatment had been performed at that time. Approximately two years later, a fluctuant swelling developed in the apical area of the teeth. Symptoms also included tenderness to percussion. Drainage was established by lingual access in the pulp chamber. Treatment was interrupted by the patient for no reason and, four years later, an attempt at apexification using Ca(OH)2 paste was carried out for six months by another dentist, but no apexification was observed for either tooth.
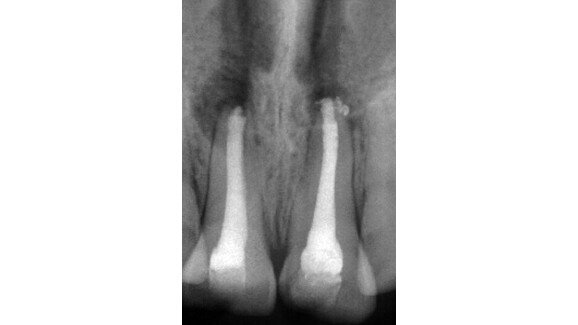
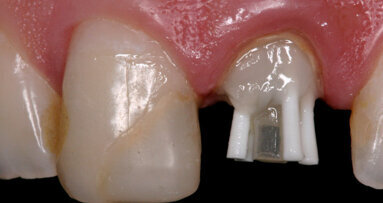
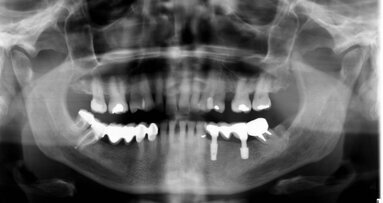
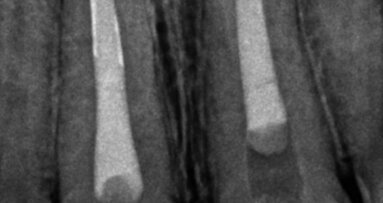
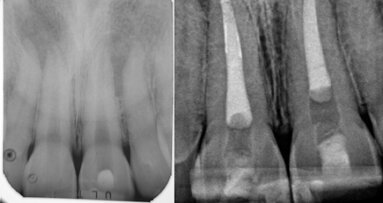
When the patient was referred to our department, extra- and intra-oral examinations (including radiology) were performed through which it was established that the left and right maxillary central incisors were in normal position with enamel–dentine fracture. The root canals were wide, the roots incompletely formed with open apices and there were periapical lesions (Fig. 1). Cleaning and shaping of the root canal system was achieved under rubber dam isolation. The solution used for irrigation was 2.5% sodium hypochlorite. Root canal length was determined using an apex locator and confirmed radiographically. Ca(OH)2 paste was placed in the canals for one week for disinfection. During the second appointment, Ca(OH)2 was removed by mechanical instrumentation and flushed from the root canals by means of sterile water irrigation. The canals were dried using sterile paper points. MTA was prepared immediately before use, placed into the canals with an MTA carrier and compacted with a hand plugger to create an apical plug of 3 to 4mm in accordance with the manufacturer’s instructions. A radiograph was taken to check whether any apical extrusion had occurred.
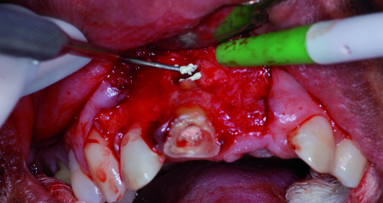
The apical plug failed in the first attempt on the right maxillary central incisor (Fig. 2). The MTA was rinsed out with sterile water and the procedure was repeated (Fig. 3). Moist paper points were placed in the canals and the access cavities were closed with a temporary restorative material, IRM (DENTSPLY).
Two days later, the coronal and middle thirds of the canals were filled with gutta-percha by a vertical warm compaction technique and the access cavities were sealed in conjunction with the final restoration (Fig. 4). Periradicular healing was assessed clinically and radiographically at six, eight and 12 months (Figs. 5 & 6). The use of MTA followed by conventional endo dontic treatment resulted in apical formation in the two central incisors (Fig. 6).
Discussion
The traditional use of Ca(OH)2 apical barriers has been associated with unpredictable apical closure, extended time taken for barrier formation, difficulties in patient compliance, and the risk of reinfection resulting from the difficulty in creating long-term seals with provisional restorations and susceptibility to root fractures arising from the presence of thin roots or prolonged exposure of the root dentine to Ca(OH)2. [7] Thus, the one-visit apexification technique is gaining popularity. One-visit apexification has been defined as the non-surgical condensation of a biocompatible material into the apical end of a root canal. The rationale is to establish an apical stop that would enable the root canal to be filled immediately. Torneck et al. [8] found that when apical closure takes place clinically with Ca(OH)2, there is incomplete bridging of the apex histologically. Periapical inflammation persists around the apices of many teeth because necrotic tissue exists in the corners and crevices of the bridge.
A major target area of biomedical research is the restoration of lost bone. To this end, a resorbable tricalcium phosphate ceramic was developed. Koenigs, Brilliant and Driskell [9] found that use of this material induced apical closure in vital teeth of primates with open apices. Regeneration of the periodontal ligament occurred around the apices of teeth and it was associated with minimal inflammatory response. Herbert documented the long-term success of using a tricalcium phosphate plug as an apical barrier for onevisit apexification. In other studies, teeth with open apices were obturated using an apical barrier with dentine and Ca(OH)2 plugs or dentine chips and hydroxyapatite. [10]
There is increasing popularity of the one-visit apexification technique using MTA as an osteoconductive apical barrier. MTA is relatively non-cytotoxic and stimulates cementogenesis. This material generates a highly alkaline aqueous environment by leaching of calcium and hydroxyl ions, rendering it bioactive by forming hydroxyapatite in the presence of phosphatecontaining fluids. Unlike the extended use of Ca(OH)2 in immature roots, prolonged filling of these roots with MTA did not reduce their fracture resistance. [11]
Torabinejad [12] reported the ingredients in MTA as tricalcium silicate, tricalcium aluminate, tricalcium oxide and silicate oxide with some other mineral oxides that were responsible for the chemical and physical properties of aggregate. The powder consists of fine hydrophilic particles that set in the presence of moisture. The hydration of the powder results in a colloidal gel with a pH of 12.5 that will set in approxi-mately 3 hours. MTA has a compressive strength equal to intermediate restorative material and SuperEBA (Bosworth) but less than that of amalgam. It is commercially available as ProRoot MTA (DENTSPLY), and has been advocated for use in the immediate obturation of an open root apex.
MTA has the ability to induce cementum-like hard tissue when used adjacent to the periradicular tissue. MTA is a promising material as a result of its superior sealing property, its ability to set in the presence of blood and its biocompatibility. Moisture contamination at the apex of tooth before barrier formation is often a problem with other materials used in apexification. As a result of its hydrophilic property, the presence of moisture does not affect its sealing ability. Shabahang et al [13] examined hard-tissue formation and inflammation histomorphologically after treating open apices in canine teeth with osteogenic protein-1, MTA and Ca(OH)2. MTA induced hard-tissue formation with the most consistency, but the amount of hard-tissue formation and inflammation was not statistically different among the three materials.
MTA has demonstrated the ability to stimulate cells to differentiate into cells that form hard tissue and to produce a hard-tissue matrix. A number of animal studies have demonstrated a more predictable healing outcome when MTA is used compared with teeth treated with Ca(OH)2. [14] In a prospective human outcome study, 57 teeth with open apices were obturated with MTA in one appointment. Forty-three of these cases were available for recall at 12 months, of which 81% of cases were classified as healed. [15] Despite its good physical and biological properties, its extended setting time has been a disadvantage. Calcium chloride has been used to stimulate the hardening process of MTA and studies have shown that both its physico-chemical properties and sealing ability were improved by the addition of calcium chloride.
Conclusions
Based on this study’s results, the following conclusions can be made:
MTA showed clinical and radiographic success as a material used to induce apical closure in necrotic immature permanent teeth. MTA is a suitable replacement for Ca(OH)2 for the apexification procedure.
A complete list of references is available from the publisher.
This article was published in roots international magazine of endodontology No. 01/2014.
Pulpal and periradicular pathology develop when the dental pulp and periradicular tissues become exposed to microorganisms. In experimental, germ-free ...
Mineral trioxide aggregate (MTA) is an endodontic sealer that emerged on the market in 1998. Through proven scientific results it has became the true ...
In recent years, dental implants have emerged as a pivotal advancement in dentistry, offering a popular solution for replacing natural teeth lost owing to ...
In recent years, orthodontists have been treating patients of increasingly younger age.1 This is due to increased attention by paediatricians, greater ...
Endodontic treatment of teeth with significant coronal destruction is a very common clinical procedure in the restorative clinical practice. When we are ...
Implant-prosthesis systems have continuously evolved regarding osseointegration, implant size reduction and long-term predictability results, guided by the ...
Non-invasive hard-tissue regeneration is not a common treatment possibility in everyday dental practice. However, in cases with extensive inflammation ...
The number of universal adhesives being offered by the dental industry nowadays can make it difficult to decide whether it may be worth switching to another...
The patient reported on in this article is a student in dentistry and his parents are both dentists. They referred their son to a good endodontist, who then...
The patient reported on in this article is a student in dentistry and his parents are both dentists. They referred their son to a good endodontist, who then...
Live webinar
Wed. 10 June 2026
11:00 am EST (New York)
Live webinar
Wed. 10 June 2026
2:00 pm EST (New York)
Nacho Fernández-Baca DDS, MSc
Live webinar
Wed. 10 June 2026
7:00 pm EST (New York)
Live webinar
Thu. 11 June 2026
1:00 pm EST (New York)
Live webinar
Thu. 11 June 2026
2:00 pm EST (New York)
Prof. Hani Ounsi DDS PhD FICD
Live webinar
Fri. 12 June 2026
8:00 am EST (New York)
Dipl.-Hdl. Joachim Brandes
Live webinar
Mon. 15 June 2026
11:00 am EST (New York)
Dr. Cristian Scognamiglio, Dr. Alessandro Perucchi



 Austria / Österreich
Austria / Österreich
 Bosnia and Herzegovina / Босна и Херцеговина
Bosnia and Herzegovina / Босна и Херцеговина
 Bulgaria / България
Bulgaria / България
 Croatia / Hrvatska
Croatia / Hrvatska
 Czech Republic & Slovakia / Česká republika & Slovensko
Czech Republic & Slovakia / Česká republika & Slovensko
 France / France
France / France
 Germany / Deutschland
Germany / Deutschland
 Greece / ΕΛΛΑΔΑ
Greece / ΕΛΛΑΔΑ
 Hungary / Hungary
Hungary / Hungary
 Italy / Italia
Italy / Italia
 Netherlands / Nederland
Netherlands / Nederland
 Nordic / Nordic
Nordic / Nordic
 Poland / Polska
Poland / Polska
 Portugal / Portugal
Portugal / Portugal
 Romania & Moldova / România & Moldova
Romania & Moldova / România & Moldova
 Slovenia / Slovenija
Slovenia / Slovenija
 Serbia & Montenegro / Србија и Црна Гора
Serbia & Montenegro / Србија и Црна Гора
 Spain / España
Spain / España
 Switzerland / Schweiz
Switzerland / Schweiz
 Turkey / Türkiye
Turkey / Türkiye
 UK & Ireland / UK & Ireland
UK & Ireland / UK & Ireland
 Brazil / Brasil
Brazil / Brasil
 Canada / Canada
Canada / Canada
 Latin America / Latinoamérica
Latin America / Latinoamérica
 USA / USA
USA / USA
 China / 中国
China / 中国
 India / भारत गणराज्य
India / भारत गणराज्य
 Pakistan / Pākistān
Pakistan / Pākistān
 Vietnam / Việt Nam
Vietnam / Việt Nam
 ASEAN / ASEAN
ASEAN / ASEAN
 Israel / מְדִינַת יִשְׂרָאֵל
Israel / מְדִינַת יִשְׂרָאֵל
 Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
 Middle East / Middle East
Middle East / Middle East



































To post a reply please login or register