Discussion
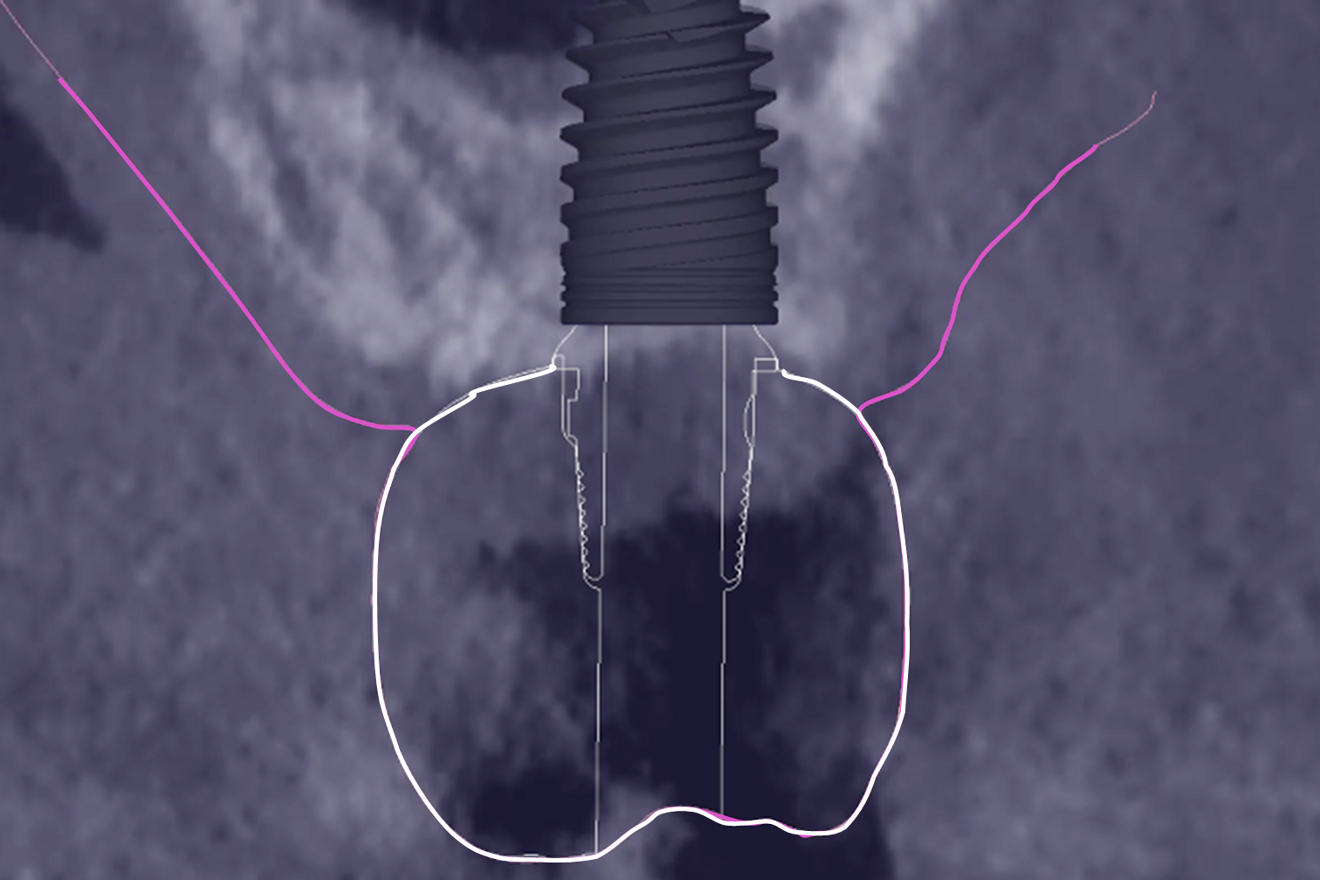
The aim of this report was to describe, through a clinical case, the type of dimensional data that it is possible to obtain from the peri-implant soft tissue with an innovative digital protocol that was very recently published16–18 and that cannot be obtained otherwise. The additional clinical effort to gain this data is minor: it requires performing three IOSs and obtaining the STL files of the various items from the manufacturer and the dental laboratory.
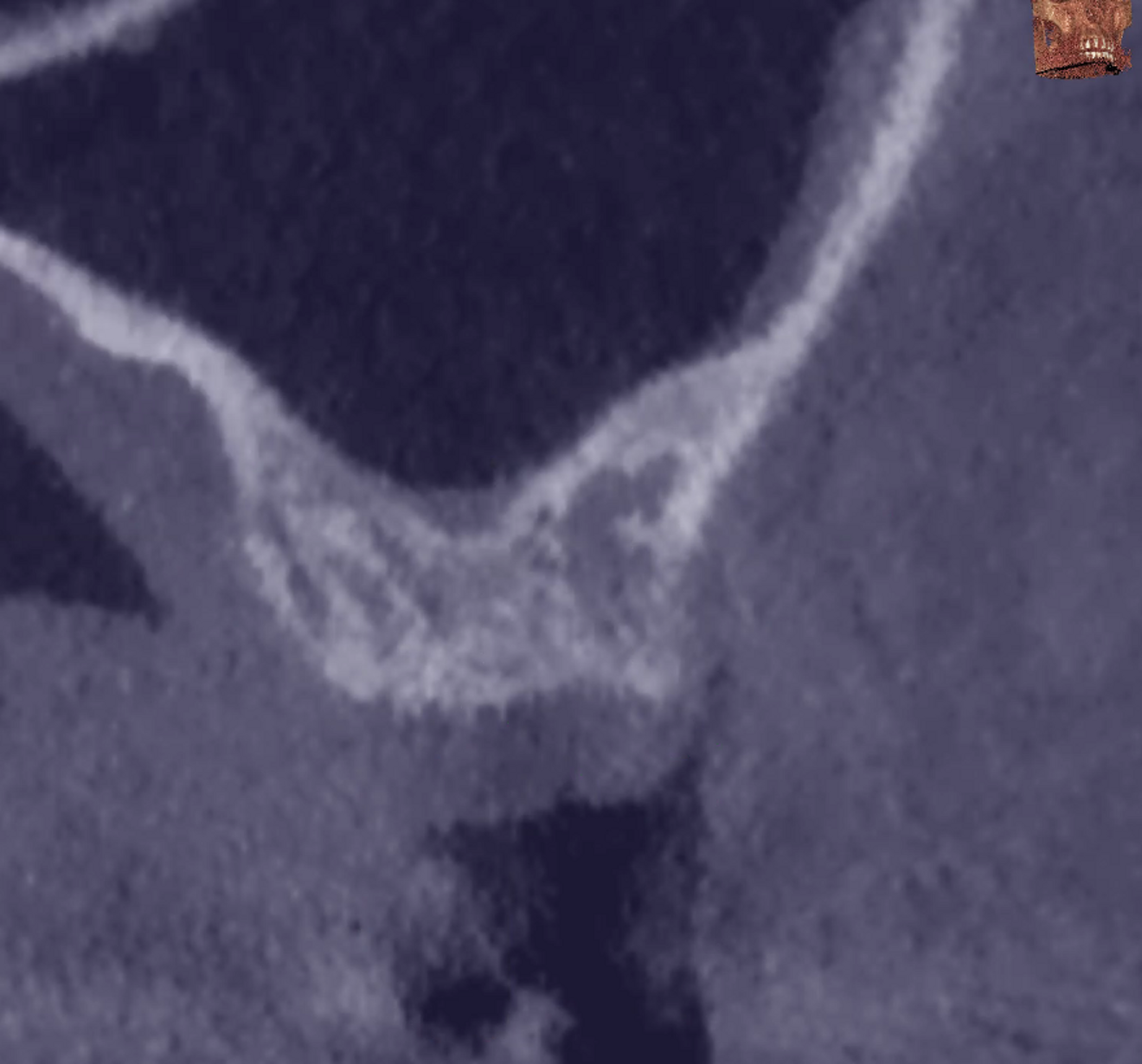
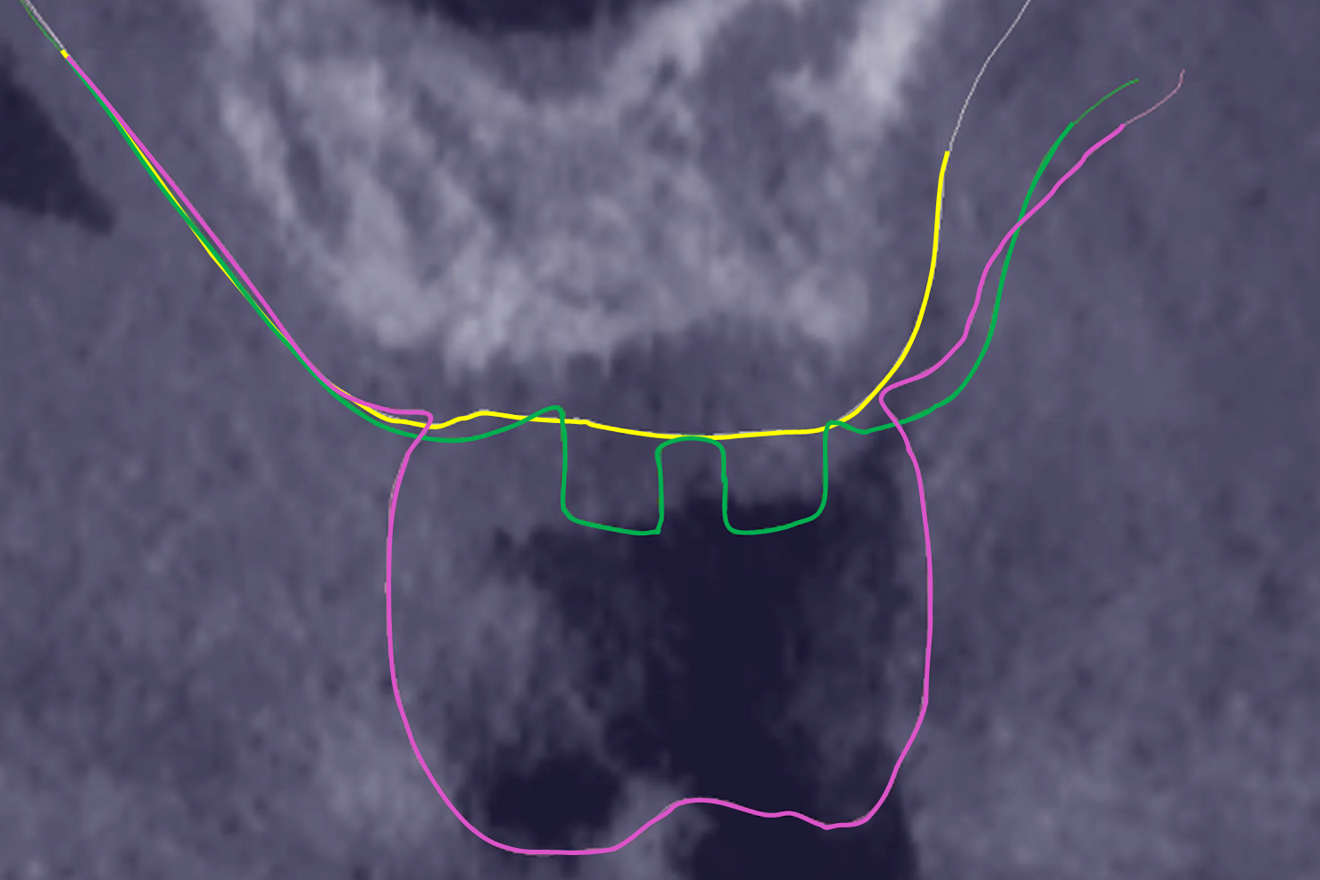
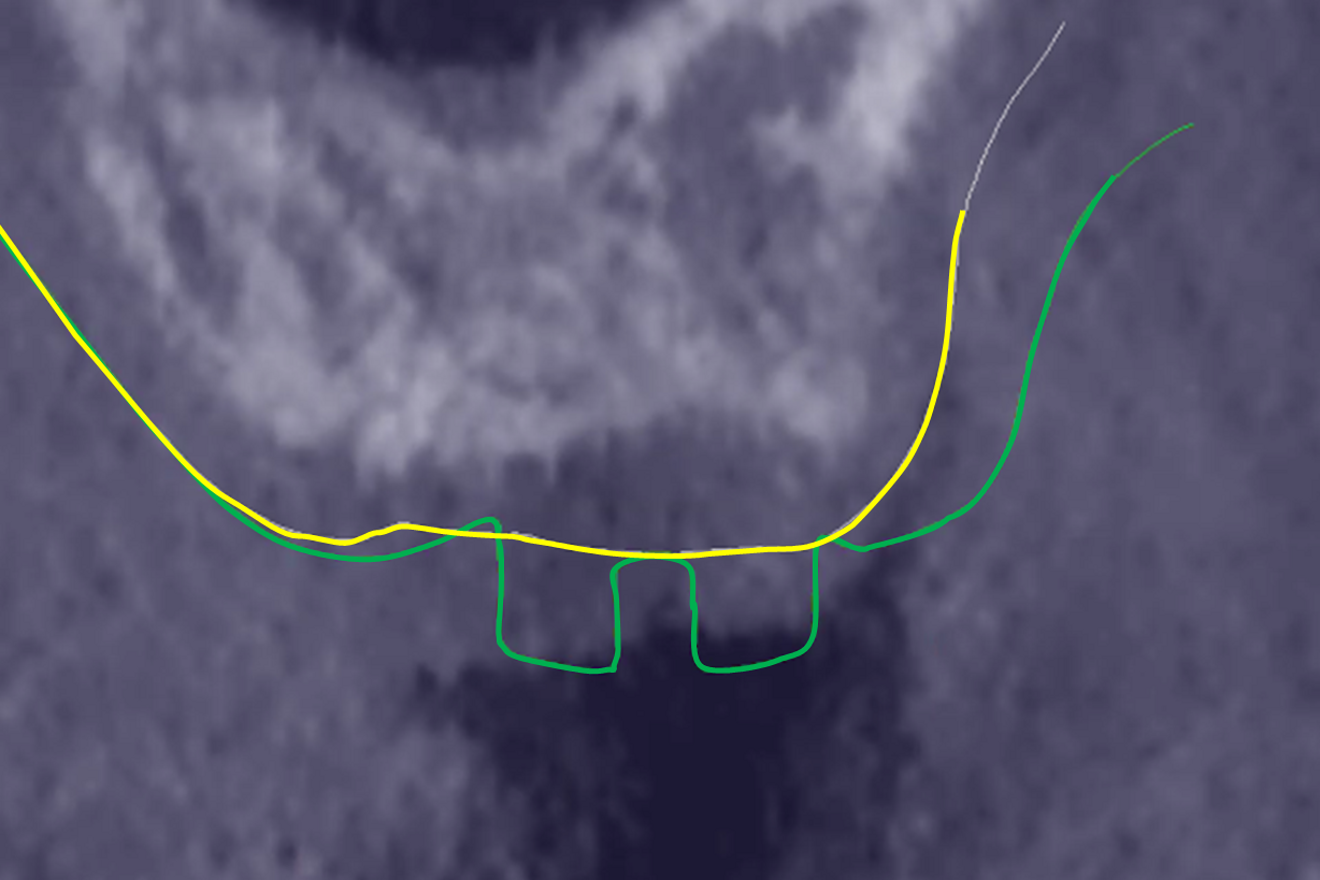
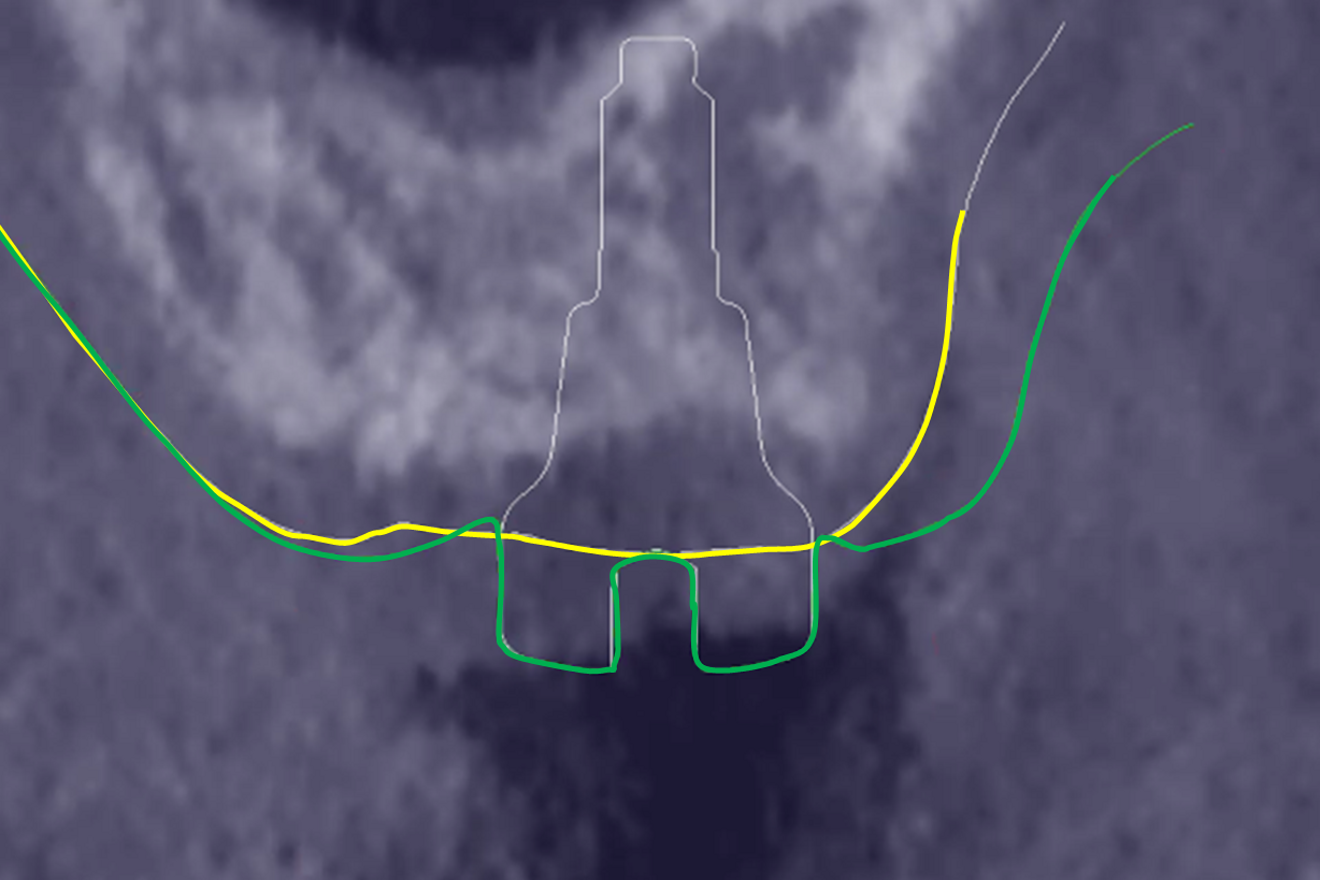
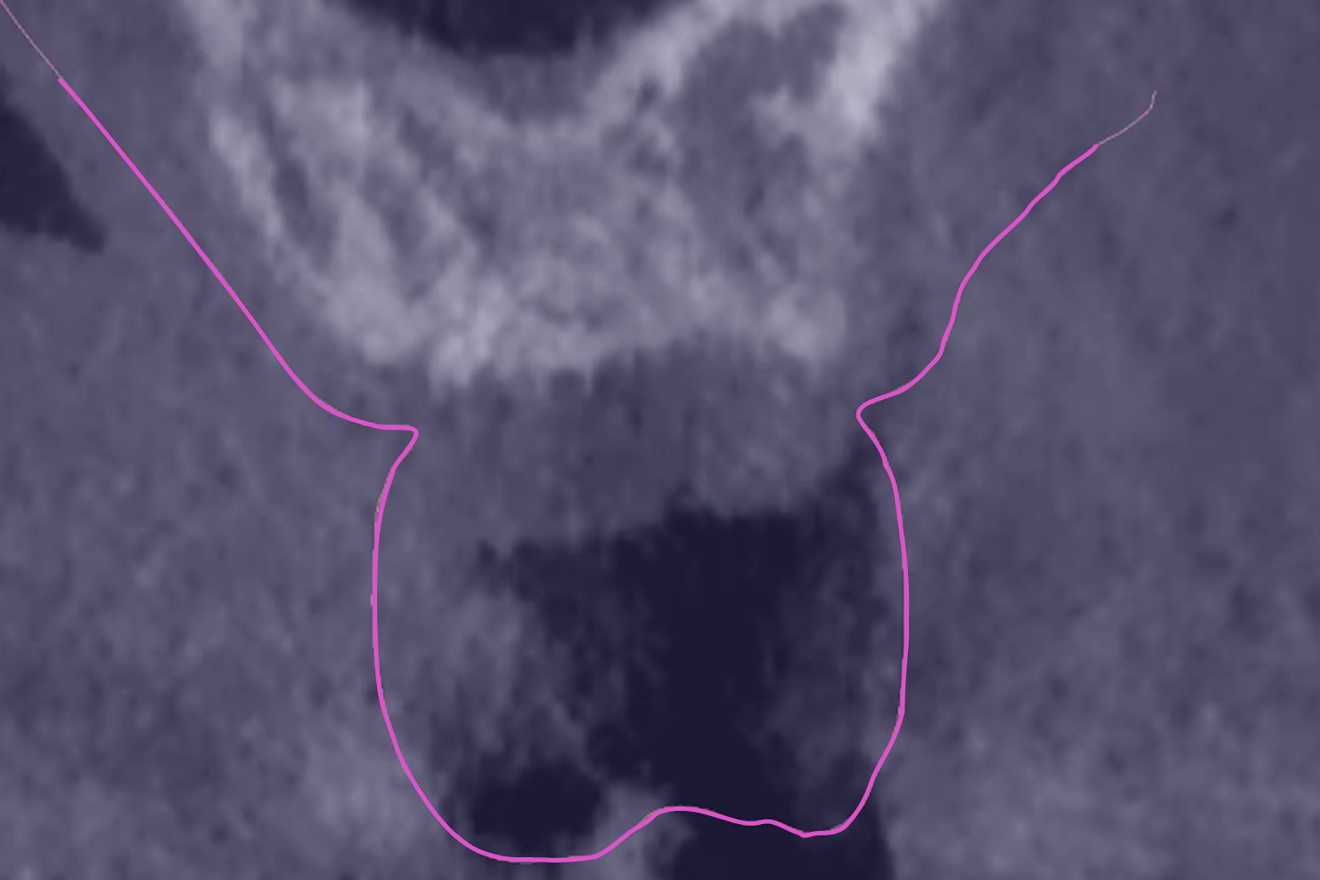
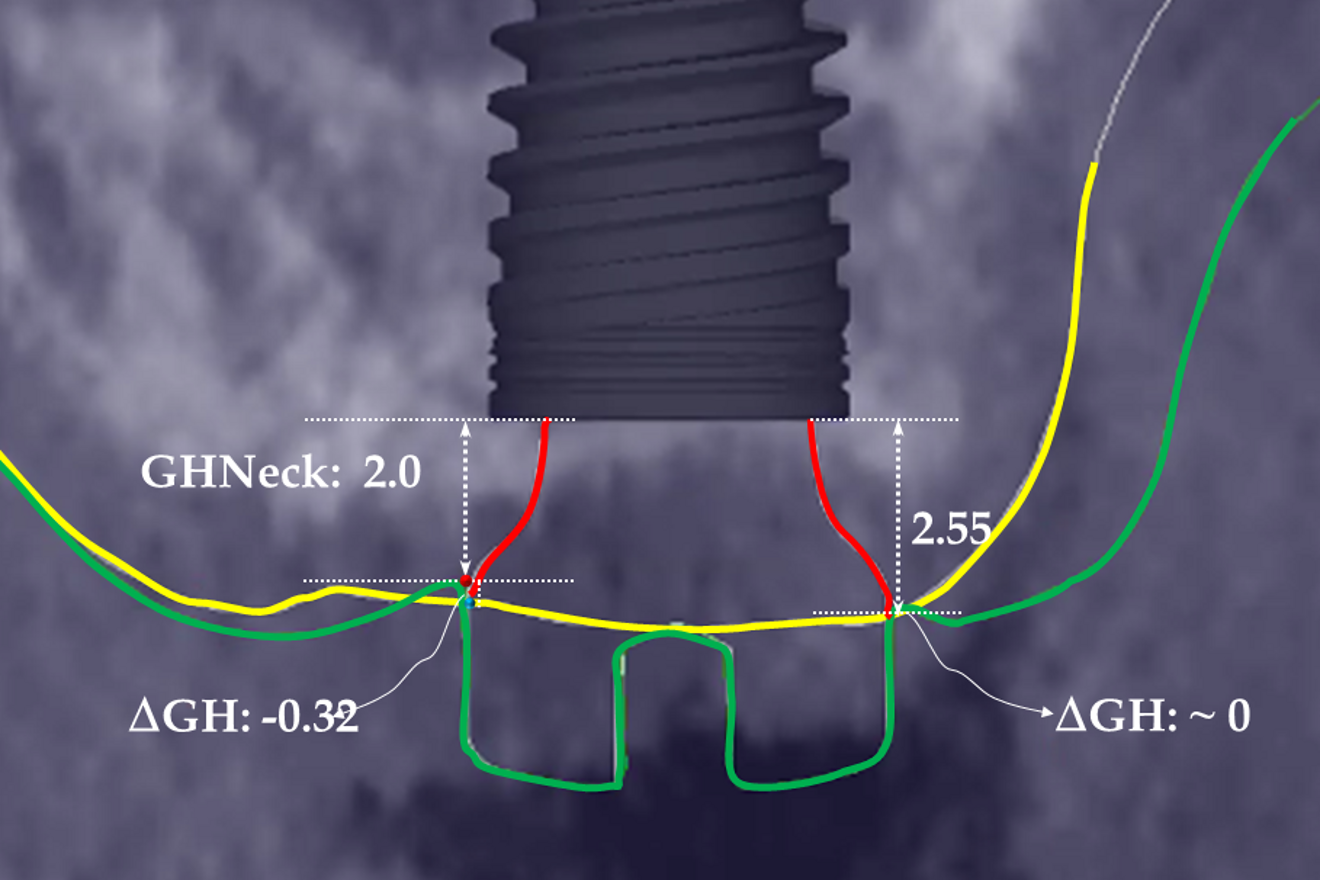
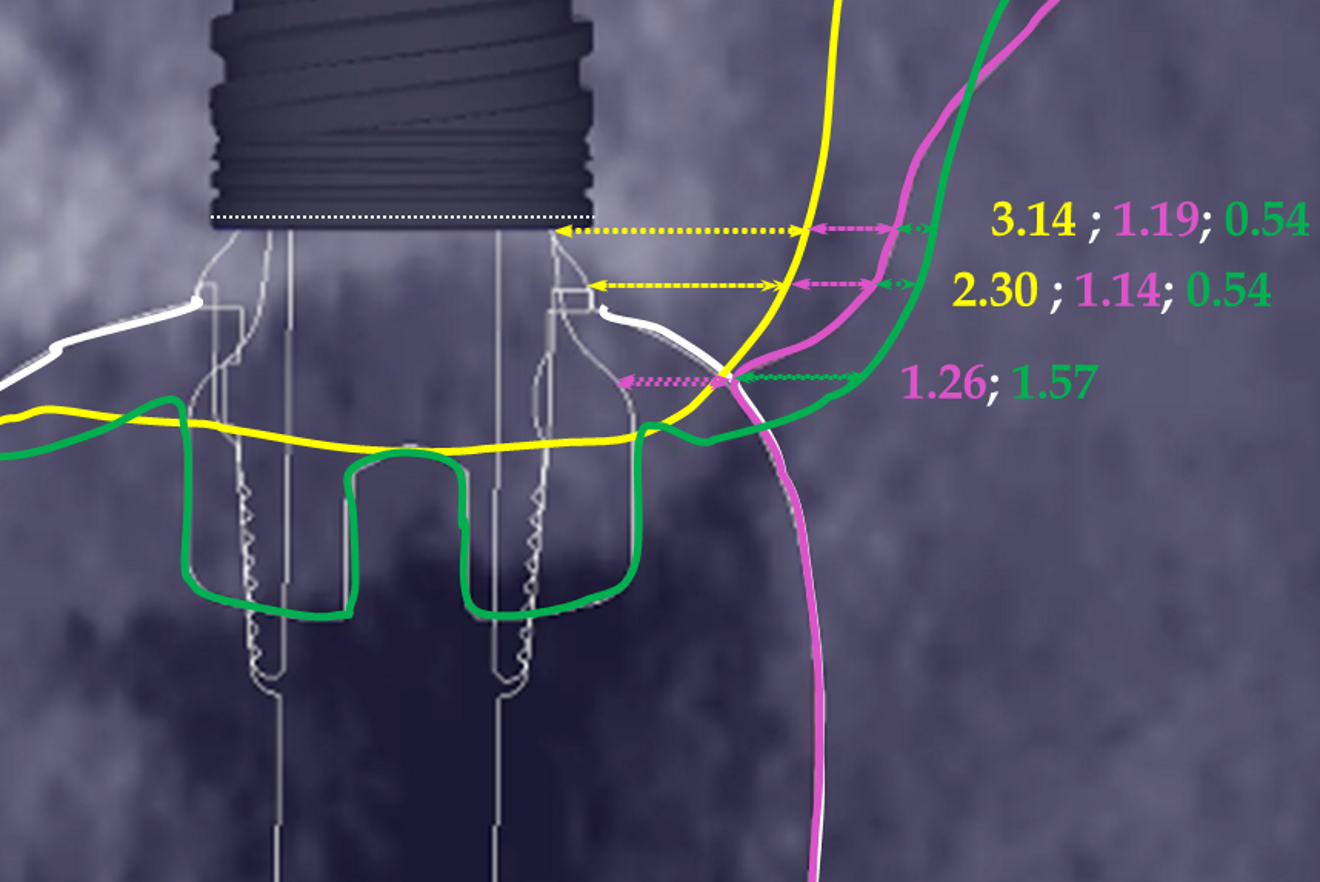
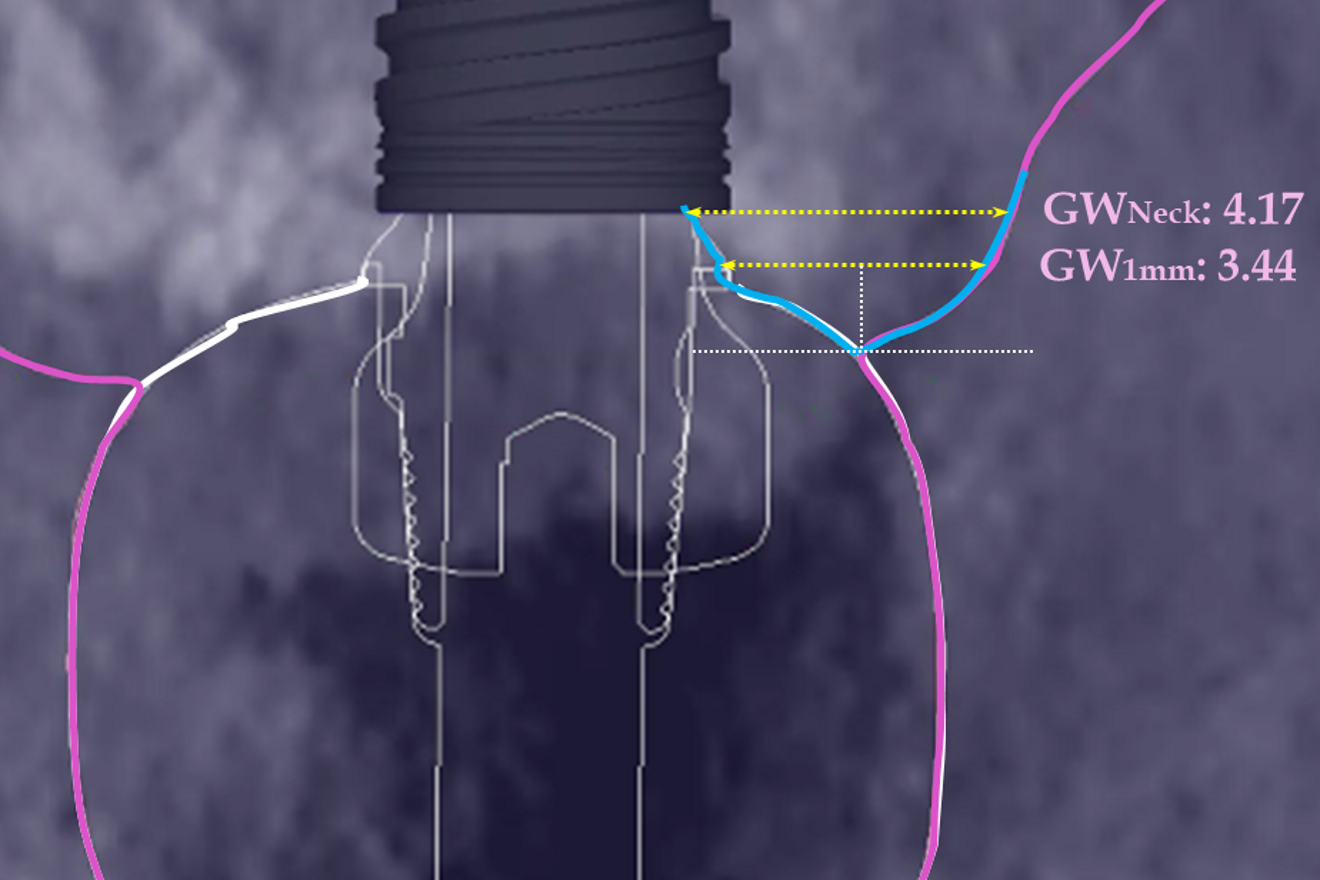
The preoperative gingival height can be read by merging IOS #0 with the CBCT scan.1–3 Determining gingival height during implant planning is important because this variable can affect how deep the implant should be placed sub-crestally without risking the implant neck being insufficiently surrounded by bone.26, 27 The routine way to measure the thickness of the gingiva is with a periodontal probe on the crest of the ridge after flap elevation;26 however, the actual place to determine the thickness is where the biological width (BW) concept applies, that is, at the emergence of the gingival margin in contact with the healing abutment. Szmukler-Moncler et al. showed that determination of the thickness at the crest does not provide an accurate estimate of the gingival thickness measured at the buccal and palatal sides of the healing abutment.28
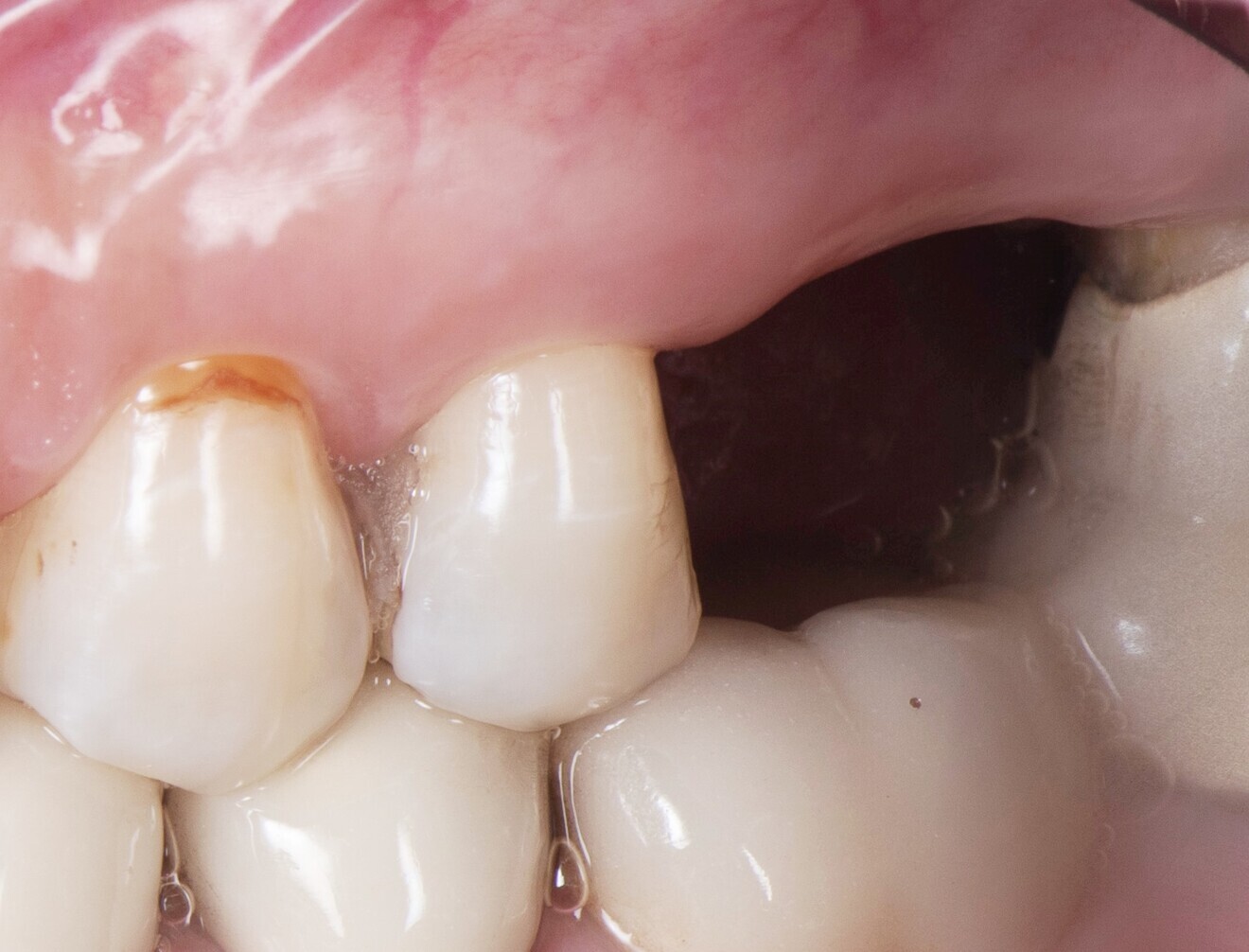
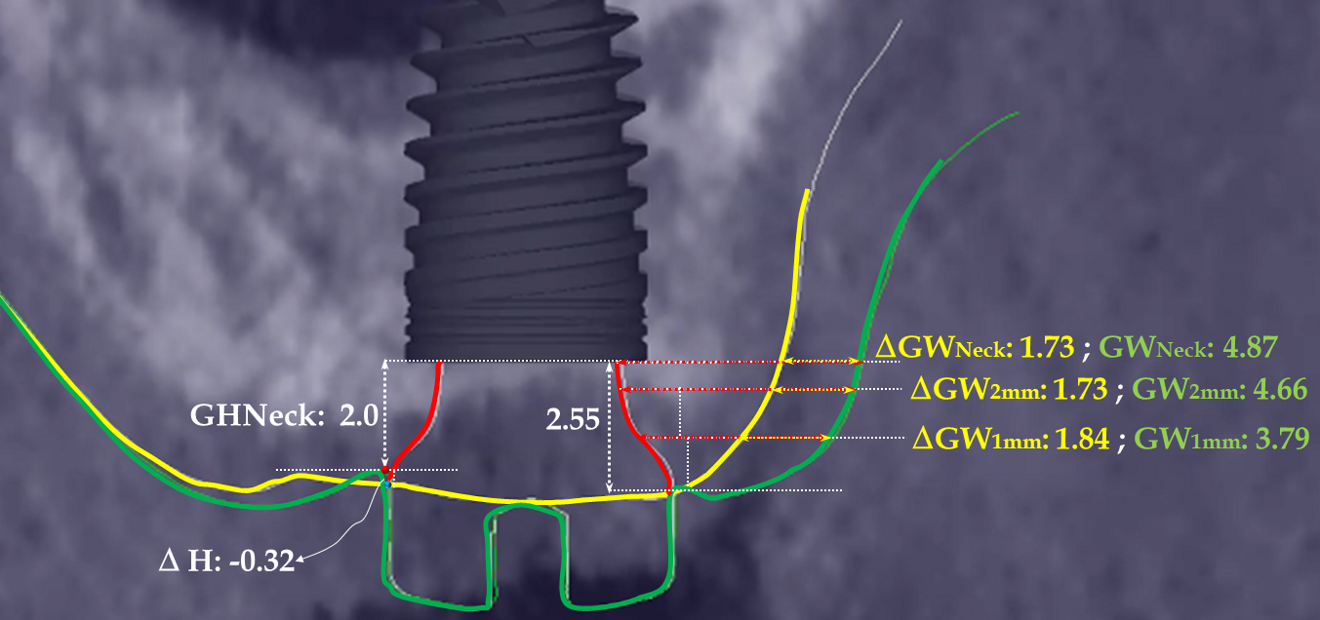
In the present case, the 2.38 mm measured at the crest suggests that the gingiva is thicker than 2 mm and might be classified as thick.26,29 However, at the place it should be measured, taking into account the biological width concept, the initial gingival heights on the buccal and palatal sides of the abutment are 1.73 mm and 1.41 mm, and therefore the gingiva should be characterised as thin. Consequently, it makes sense to anticipate crestal bone loss down to at least the implant neck in order to achieve acceptable dimensions of the biological width.
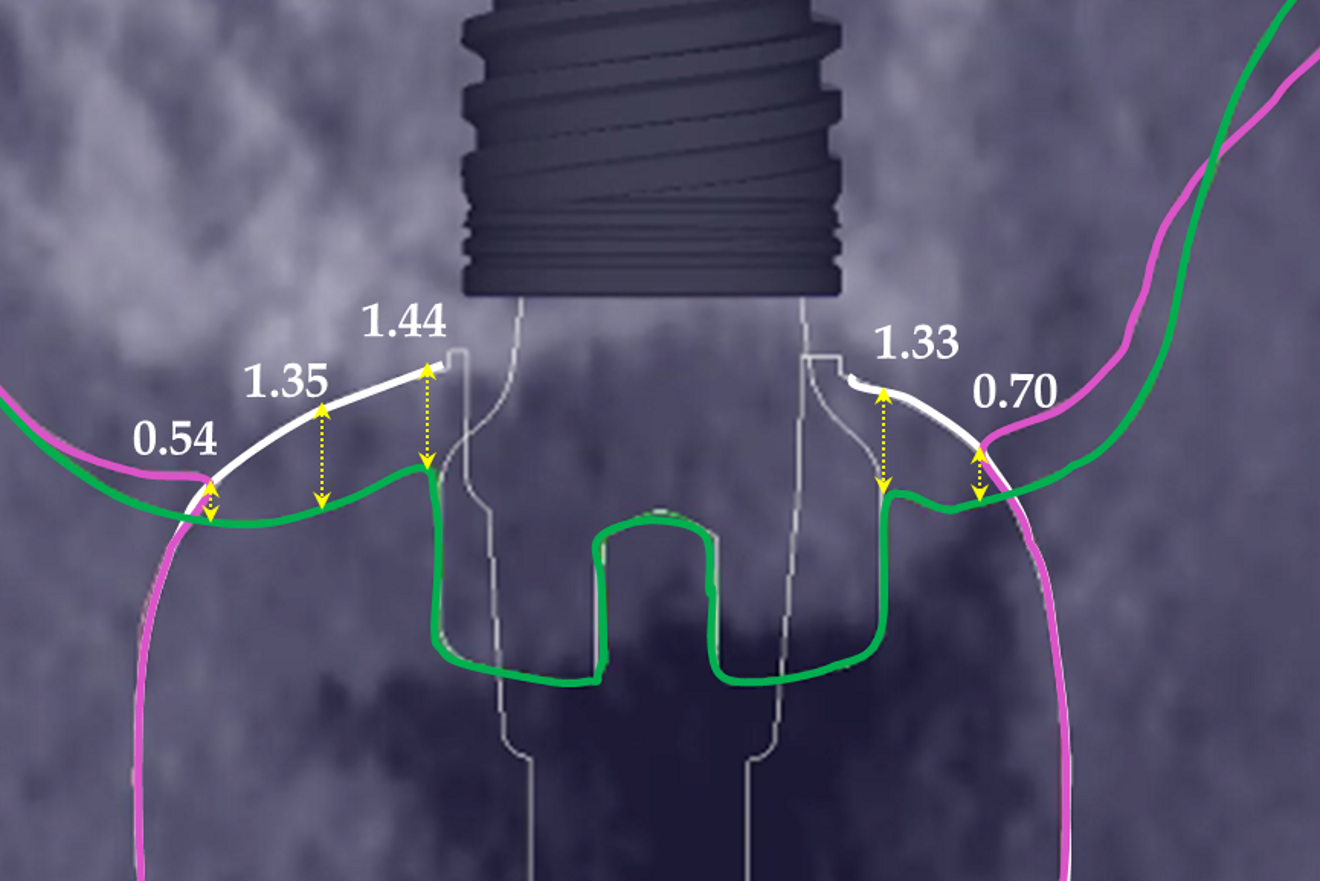
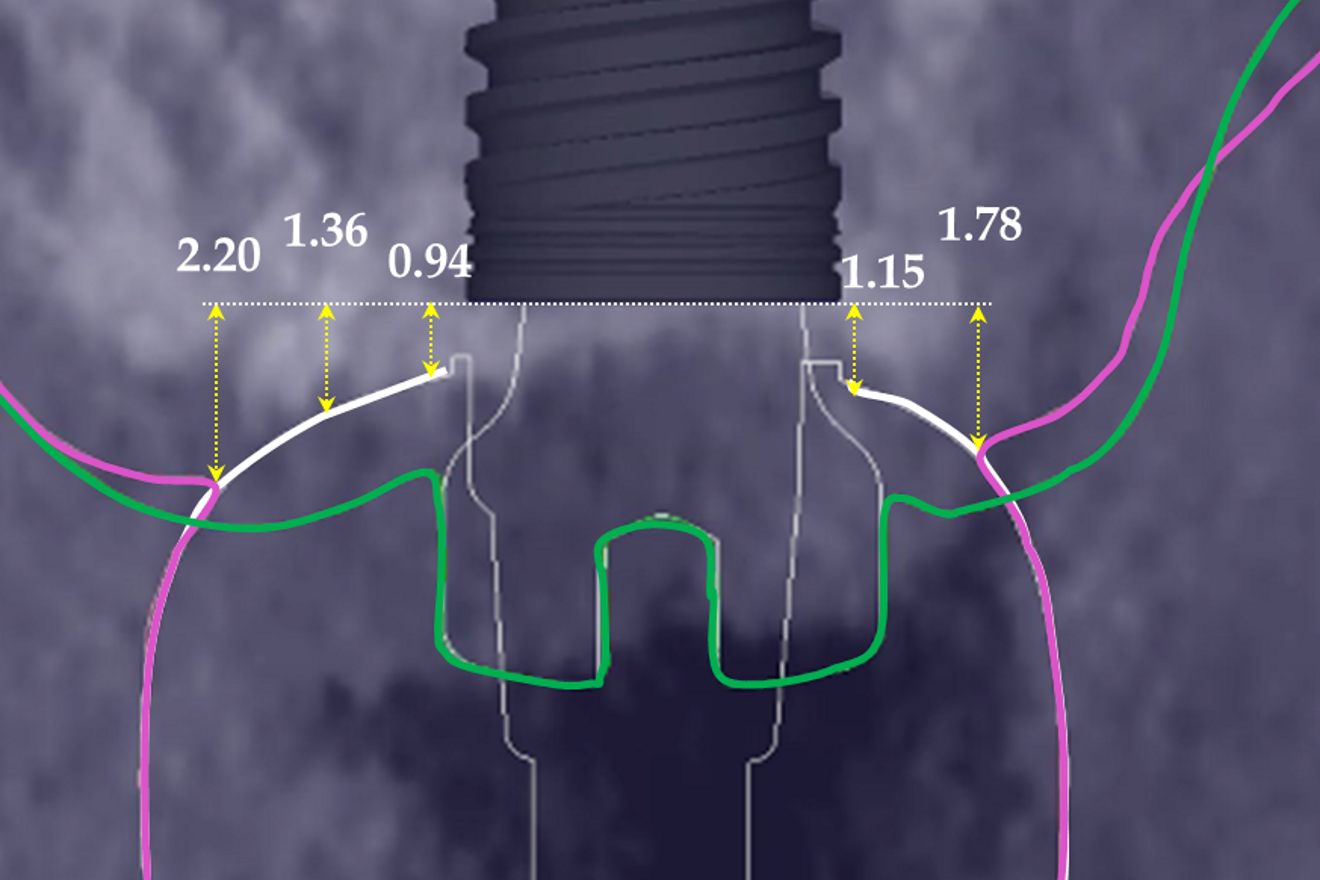
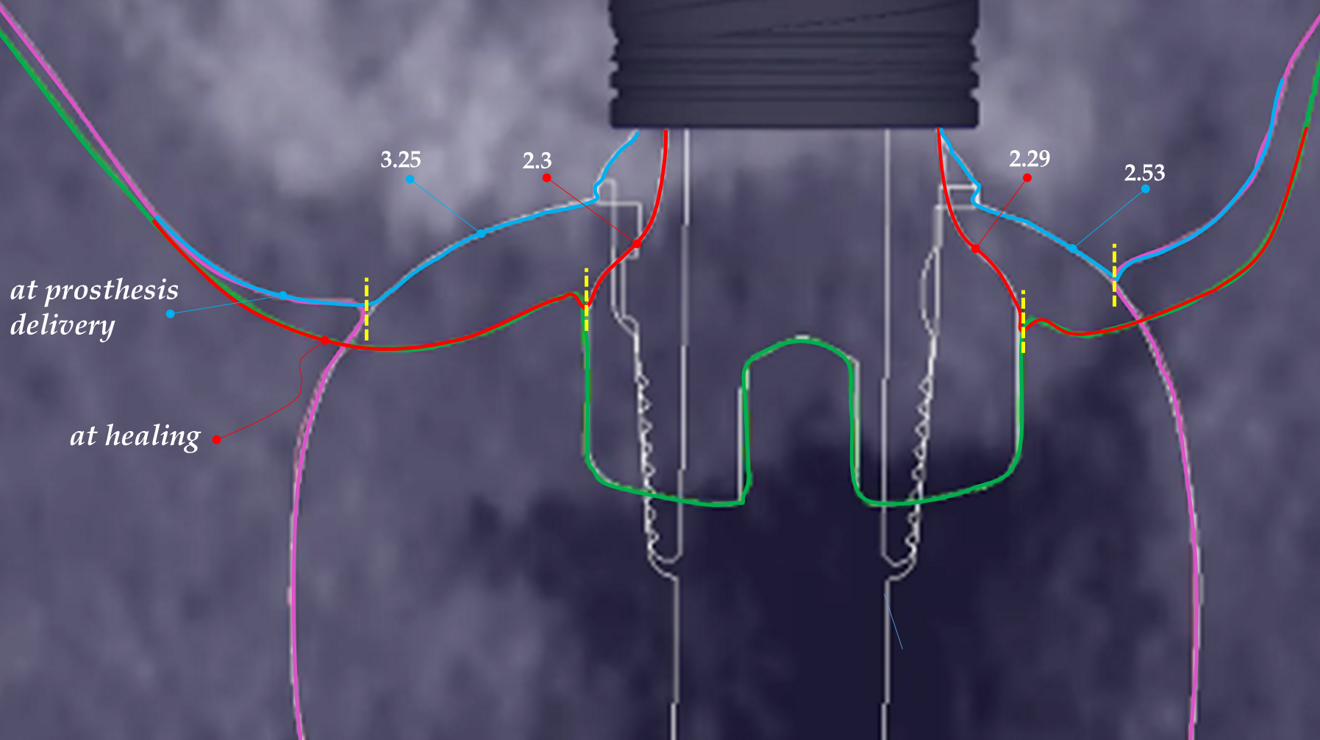
At the end of the healing period, it appeared that the modified roll technique did not affect the gingival height; however, it did increase the width of the buccal gingiva significantly. The gain in width was 1.84 mm and 1.73 mm at 1.0 mm and 2.0 mm below the gingival margin, respectively, and the overall width reached 4.66 mm and 4.87 mm. Today, this digital protocol is the only way to access these clinical variables in a non-invasive way. Placement of the crown instantly and substantially modified the gingival height by 0.70–1.33 mm on the buccal side and by 0.54–1.44 mm on the palatal side. Pressure on the gingiva was clinically evidenced by bleaching of the buccal and palatal gingiva. Again, only this digital methodology is able to provide such a precise insight into the resulting dimensional changes. One can legitimately assume that this compression of the gingiva might lead to a subsequent rearrangement between the compressed soft tissue and the underlying crestal bone and in turn to bone resorption. After delivery of the crown, the digital superimposition showed that part of the width gained by the end of soft-tissue healing by the time-consuming modified palatal roll technique had vanished.
Conclusion
The inventive step of the present digital methodology consists of merely merging the IOSs that are typically obtained 6,7 with the STL files of the various implant-related items.16-18,28 This single addition brings about an illuminating difference, as it renders the gingiva transparent, enabling reading of the vertical and horizontal dimensions of the peri-implant gingiva and their changes at every level of the abutment and at every stage of the treatment. This is otherwise unattainable in clinical research.
Implementation of this digital workflow in clinical research will help refine the data acquired from protocols and techniques of soft-tissue thickening, providing information on not only the tissue thickness that has been gained but also the entire thickness of the gingiva.



 Austria / Österreich
Austria / Österreich
 Bosnia and Herzegovina / Босна и Херцеговина
Bosnia and Herzegovina / Босна и Херцеговина
 Bulgaria / България
Bulgaria / България
 Croatia / Hrvatska
Croatia / Hrvatska
 Czech Republic & Slovakia / Česká republika & Slovensko
Czech Republic & Slovakia / Česká republika & Slovensko
 France / France
France / France
 Germany / Deutschland
Germany / Deutschland
 Greece / ΕΛΛΑΔΑ
Greece / ΕΛΛΑΔΑ
 Hungary / Hungary
Hungary / Hungary
 Italy / Italia
Italy / Italia
 Netherlands / Nederland
Netherlands / Nederland
 Nordic / Nordic
Nordic / Nordic
 Poland / Polska
Poland / Polska
 Portugal / Portugal
Portugal / Portugal
 Romania & Moldova / România & Moldova
Romania & Moldova / România & Moldova
 Slovenia / Slovenija
Slovenia / Slovenija
 Serbia & Montenegro / Србија и Црна Гора
Serbia & Montenegro / Србија и Црна Гора
 Spain / España
Spain / España
 Switzerland / Schweiz
Switzerland / Schweiz
 Turkey / Türkiye
Turkey / Türkiye
 UK & Ireland / UK & Ireland
UK & Ireland / UK & Ireland
 Brazil / Brasil
Brazil / Brasil
 Canada / Canada
Canada / Canada
 Latin America / Latinoamérica
Latin America / Latinoamérica
 USA / USA
USA / USA
 China / 中国
China / 中国
 India / भारत गणराज्य
India / भारत गणराज्य
 Pakistan / Pākistān
Pakistan / Pākistān
 Vietnam / Việt Nam
Vietnam / Việt Nam
 ASEAN / ASEAN
ASEAN / ASEAN
 Israel / מְדִינַת יִשְׂרָאֵל
Israel / מְדִינַת יִשְׂרָאֵל
 Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
Algeria, Morocco & Tunisia / الجزائر والمغرب وتونس
 Middle East / Middle East
Middle East / Middle East










































To post a reply please login or register